
Daniel Colón-Ramos, Ph.D.
Associate Professor
Program in Cellular Neuroscience, Neurodegeneration and Repair, Department of Cell Biology
Department of Neuroscience
Yale University School of Medicine
Serving the community as a scientist
As a Puerto Rican, it has always been very important to me that my work benefit my community. Although I was interested in science from a young age, as it came time for me to start making career decisions, I couldn’t see how, as a scientist, I could positively impact my community. In part, this was because I was not sure what it meant to be a scientist. I initially gravitated toward a career with direct applications to health in my community, such as medicine or social justice work. However, over the course of my journey, I came to realize that the best way to give back was to follow my passion and use my skills, knowledge, and ties to benefit communities that are underserved in the sciences.
One of the big turning points in my career came after college. During college, I merged my interest in science with my interest in social justice by working on ethnobotanical projects such as studying the use of medicinal plants by indigenous communities in Central America. But my mind always returned to the basic science; I itched to know how these medicinal plants were actually working. Although I had an opportunity to pursue a Fulbright to continue similar work after college, I was still unsure. I didn’t have the training to do the work as effectively as I wanted to, and I felt I wasn’t taking advantage of my intellectual interests, knowledge, and connections.
Around this time, I reached out to a Puerto Rican scientist, Mariano García-Blanco, who had graduated from my high school in Puerto Rico (Colegio San Ignacio) and was a professor at Duke University. We had a long conversation about my career trajectory, plans, and concerns. He told me that while he could not answer my questions, he could offer me the opportunity to work in his lab at Duke University for a year to experience full time research and figure out the answers for myself.
I joined Dr. García-Blanco’s lab. We applied for, and received, an NIH diversity supplement to his R01 to use the algae Chlamydomonas reinhardtii to study how nuclear architecture changes during upregulation of transcription —establishing a new experimental system in the lab. Thanks to the diversity supplement, I was able to work as an independent investigator rather than a technician. I used the funding to visit Rob Singer's lab at Albert Einstein College of Medicine and jointly developed a new protocol that allowed fluorescent in situ hybridization (FISH) inChlamydomonas. That year took advantage of the NIH grant-writing workshops, went to my first scientific meeting, and published my first paper in Developmental Cell.
This experience cemented my desire to continue in science. I decided to enter the Ph.D. program at Duke, and joined the lab of Sally Kornbluth. From Dr. Kornbluth, I learned how to identify important research questions, design experiments, and structure the work for publication. I ended up seeking out and establishing a lot of collaborations during graduate school while pursuing my research questions of interest. If I had to define one thing that helped me during those early experiences, was to seek out advice, and learn, from the best.
I then worked as a postdoctoral fellow at Stanford University with Kang Shen, another superb mentor who helped me transition into a role as an independent investigator. After graduate school, I felt I could solve research problems, but I hadn’t yet conceptualized the fundamental questions I would base my career on. The process of applying for the K99/R00 from NINDS pushed me to think beyond the paper into what I wanted my longer term contributions to be as a scientist. It also gave me the opportunity to get feedback from the community on the proposed projects.
The K99/R00 funding let me focus on what I needed to do as a postdoctoral researcher to promote my career without having to compete with the demands of the research my advisor was funded to do. It also ensured that I develop, and continue, an independent project to start my own lab. Finally, as a new faculty member, I could focus on my research and not worry so much about funding during those initial, critical transition years.
Over the course of my career, I have come to realize that scientists occupy very powerful positions in terms of knowledge creation, distribution of knowledge, and training. Contrary to what I feared as an undergraduate, I have found many ways to impact communities that are underserved in the sciences. I have worked to increase access to science through initiatives like Ciencia Puerto Rico, a non-profit organization that promotes scientific research and education in Puerto Rico.
In my own lab, we value diversity because it benefits the science to include different perspectives in our work. So far, I have mentored two students—Luis Martinez Velazquez and Lucelenie Rodriguez Laureano—who have also taken advantage of NINDS diversity supplements to launch their careers and to push important lab projects forward. NINDS funding mechanisms have now benefited the next generation of scientists, and continue to benefit my lab and our research.
Current Research
A central unsolved question in neurobiology is how synapses are precisely assembled to build the neuronal architecture that underlies behavior. To address this, we have developed tools in the thermotaxis circuit of the nematode worm, C. elegans. The system we developed enables unbiased genetic screens to identify novel pathways that instruct synaptogenesis in vivo, and single-cell manipulation of these pathways to understand how they influence behavior. Our findings serve as a foundation for the next phase of our research: integrating multidisciplinary approaches in single cells and circuits to investigate the relatively unexplored interplay between the cell biology of the synapse and behavior. As mechanisms underlying synapse structure and function are conserved, our research program could enhance our understanding of synaptic cell biology in higher organisms, which may be important for disease.
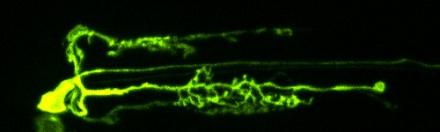
In the nematode worm C. elegans, the main serotonergic neuron, called NSM, elaborates axonal arbors within a precise neuroanatomical location overlying the nerve ring (Axang et al., 2008). We recently developed a system that allows us to study the growth of NSM and its synapses in vivo, in real time and with single cell resolution. This gives us an unprecedented opportunity to dissect the cellular and molecular mechanisms that regulate serotonergic synapse development in vivo.
