
Catrina Sims-Robinson, Ph.D.
Assistant Professor
Department of Neurology
Department of Neuroscience
Medical University of South Carolina
Embracing opportunities for growth to become a better scientist
My mother was initially diagnosed with breast cancer when I was 8-years-old, and again when I was 13-years-old. Witnessing her undergo multiple surgeries, radiation, and chemotherapy sparked my initial interest in science and the medical field, and my dream became to go to medical school. Given that medical school was my goal, I participated in various STEM-related programs at Tuskegee University throughout my time in middle and high school. Furthermore, every decision in college, from the choice of school, to the selected major, to the extracurricular activities were carefully chosen to ensure that I attained my goal.
I faced my first obstacle, however, in the form of a biochemistry course I took my junior year of college. I struggled with the course, but I found a tutor who was a Ph.D. graduate student at the time. My tutor, despite my ignorance of biochemistry, thought I asked insightful questions and suggested I meet with her advisor to discuss the possibility of working in the laboratory. As a result, I began to volunteer in a neurophysiology laboratory. I simultaneously worked with my tutor on her project, which focused on understanding the mechanisms that contributed to an increased risk of Alzheimer’s disease with diabetes. I was fascinated by this project because my grandmother, who was a diabetic, had also been diagnosed with dementia.
I graduated with a Bachelor’s of Science in Laboratory Technology and began the arduous process of applying to medical school. Amid medical school applications, my undergraduate research mentor suggested that I instead enroll in graduate school and pursue a Master’s degree. Initially, I thought, “A Master’s degree would look great on my medical school application.” I naively thought the only way I could contribute to science was by being a medical doctor, so my mind was still set on medical school.
To further complicate my path toward a position in the healthcare field, a year into the program for a Master’s degree my advisor asked me if I would be interested in pursuing a Ph.D. in Pharmaceutical Sciences instead of attending medical school. I never thought of research as a career, though after talking to my advisor, I understood that being a medical doctor was not the only way I could make an impact in the fields of science and healthcare. I realized that I truly had a passion for research and that this was the path I should follow.
One of the challenges I still had to overcome as I sought my Ph.D., though, was my inability to see the value of criticism. As an African American woman, I felt that I consistently needed to have the answer while portraying confidence and strength. Criticism initially felt like an affront and exposed my weaknesses. I quickly learned that embracing criticism, and listening to other peoples’ ideas and opinions, were actually opportunities for growth and would ultimately make me a better scientist.
After obtaining my Ph.D., I completed a postdoctoral fellowship at the University of Michigan. While I was successful at obtaining training grant awards, my grant applications for individual fellowship awards were not successful. I transitioned into a junior faculty position at the University of Michigan and began my journey to funding. My postdoctoral mentor informed me about the NINDS diversity faculty K01 award. My first K01 submission was ‘Not Discussed’ and I was completely discouraged. Another faculty member told me, “I don’t want to discourage you, but it is nearly impossible for an application to go from Not Discussed to Funded”.
I had to remember that criticism was my opportunity to improve. This was the first time I realized that to be successful, I had to evolve my way of thinking, re-evaluate my plans, take advantage of available resources, and be resilient. I therefore talked with my program officer, worked with my university to assemble a panel of faculty members who would provide an internal review of my resubmission, attended a grant writing workshop, and participated in the NINDS sponsored Grant Writing Workshop for Diverse Researchers.
I was then awarded a diversity K01, which subsequently laid the foundation for my research career. The data generated from this K01 was instrumental for my funded R01 application. Overall, through my experiences thus far, I understand the importance of having great mentors. I have been fortunate to have one mentor in particular who has been, and still is, my advocate. She has always been brutally honest about hardships that I may face in academia as not only a woman, but as an African American woman. I encourage trainees in the beginning of their careers to realize that successes are just as important as failures. One will never succeed without trying, and one will never know weaknesses without failure.
Current Research
My lab focuses on understanding the impact of metabolic disorders on both normal and pathological brain aging. My lab is particularly interested in the impact of diet-induced metabolic risk factors on cognitive function, Alzheimer’s disease onset and progression, and stroke recovery. Metabolic risk factors such diabetes, obesity, insulin resistance, and high cholesterol alters brain function and has negative consequences on learning and memory. These factors likely induce changes in various pathways years prior to any noticeable symptom. Hence, the goal of my laboratory is to identify these early changes in order to develop novel therapeutic targets to delay or prevent disease progression. One pathway that we study is altered insulin signaling.
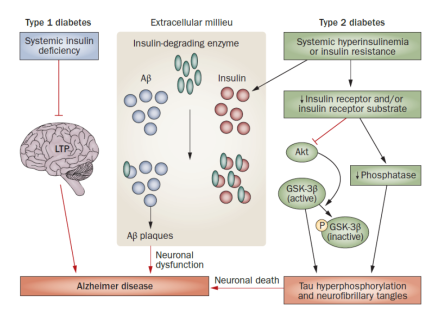
Altered insulin signaling in diabetes might contribute to Alzheimer disease pathophysiology. In type 1 diabetes insulin deficiency attenuates LTP and might lead to deficits in spatial learning and memory. In type 2 diabetes, insulin resistance leads to both Aβ plaque formation and tau hyperphosphorylation. During hyperinsulinemia, insulin and Aβ compete for insulin-degrading enzyme leading to Aβ accumulation and plaque formation. A decrease in insulin receptor signaling leads to inhibition of Akt and dephosphorylation (activation) of GSK-3β, and results in tau hyperphosphorylation. Abbreviations: Aβ, amyloid-β; GSK-3β, glycogen synthase kinase 3β; LTP, long-term potentiation; P, phosphate. (Sims-Robinson et al., Nature Reviews Neurology, 6, 551-559, 2010. PMCID: PMC3199576)
