- Organization Chart
- Appropriation Language
- Amounts Available for Obligation
- Budget Graphs
- Authorizing Legislation
- Appropriations History
- Justification of Budget Request
- Detail of Full-Time Equivalent Employment (FTE)
- Detail of Positions
Organization Chart
National Institutes of Health
National Institute of Neurological Disorders and Stroke
For carrying out section 301 and title IV of the PHS Act with respect to neurological disorders and stroke, $1,312,998,000.
| Source of Funding | FY 2016 Actual |
FY 2017 Annualized CR | FY 2018 President's Budget2 |
|---|---|---|---|
| Appropriation | $1,696,139 | $1,696,139 | $1,355,998 |
| Mandatory Appropriation: (non-add) | |||
| Type 1 Diabetes | (0) | (0) | (0) |
| Other Mandatory financing | 0 | (0) | (0) |
| Rescission | 0 | -3,224 | 0 |
| Sequestration | 0 | 0 | 0 |
| Zika Intra-NIH Transfer | -2,348 | 0 | 0 |
| Subtotal, adjusted appropriation OAR HIV/AIDS Transfers |
$1,693,791 -959 |
$1,692,915 0 |
$1,355,998 0 |
| Subtotal, adjusted budget authority Unobligated balance, start of year Unobligated balance, end of year |
$1,692,832 0 0 |
$1,692,915 0 0 |
$1,355,998 0 0 |
| Subtotal, adjusted budget authority Unobligated balance lapsing |
$1,692,832 -2 |
$1,692,915 0 |
$1,355,998 0 |
| Total obligations | $1,692,830 | $1,692,915 | $1,355,998 |
1 Excludes the following amounts for reimbursable activities carried out by this account:
FY 2016 - $20,596 FY 2017 - $20,670 FY 2017 - $19,530
2 Includes $43 million derived bv transfer from the NIH Innovation Account
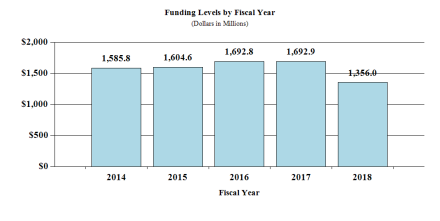
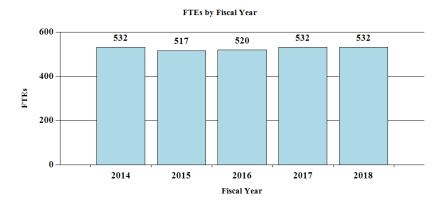
Fiscal Year 2018 Budget Graphs
History of Budget Authority and FTE's:
Authorizing Legislation
| PHS Act/ Other Citation |
U.S. Code Citation |
2017 Amount Authorized |
F Y 2017 Annualized CR |
2018 Amount Authorized |
F Y 2018 PB |
|
|---|---|---|---|---|---|---|
| Research and Investigation | Section 301 | 42§241 | Indefinite | $1,692,915,000 | Indefinite | $1,355,998,000 |
| National Institute of Neurological Disorders and Stroke |
Section 401(a) | 42§281 | Indefinite | Indefinite | ||
| Total, Budget Authority | $1,692,915,000 | $1,355,998,000 |
Authorizing Legislation
| Fiscal Year | Budget Estimate to Congress | House Allowance | Senate Allowance | Appropriation |
|---|---|---|---|---|
| 2008 | $1,537,019,000 | $1,559,106,000 | $1,573,268,000 | $1,571,353,000 |
| Rescission | $27,452,000 | |||
| 2009 | $1,545,397,000 | $1,598,521,000 | $1,588,405,000 | $1,593,344,000 |
| Rescission | $0 | |||
| Supplemental | $8,212,000 | |||
| 2010 | $1,612,745,000 | $1,650,253,000 | $1,620,494,000 | $1,636,371,000 |
| Rescission | $0 | |||
| 2011 | $1,681,333,000 | $1,678,696,000 | $1,636,371,000 | |
| Rescission | $14,368,312 | |||
| 2012 | $1,664,253,000 | $1,664,253,000 | $1,603,741,000 | $1,629,445,000 |
| Rescission | $3,079,651 | |||
| 2013 | $1,624,707,000 | $1,629,631,000 | $1,626,365,349 | |
| Rescission | $3,252,731 | |||
| Sequestration | $81,632,357 | |||
| 2014 | $1,642,619,000 | $1,631,703,000 | $1,587,982,000 | |
| Rescission | $0 | |||
| 2015 | $1,608,461,000 | $1,605,205,000 | ||
| Rescission | $0 | |||
| 2016 | $1,660,375,000 | $1,656,758,000 | $1,694,758,000 | $1,696,139,000 |
| Rescission | $0 | |||
| 20171 | $1,695,180,000 | $1,751,049,000 | $1,803,306,000 | $1,696,139,000 |
| Rescission | $3,224,000 | |||
| 2018 | $1,355,998 |
1 Budget Estimates to Congress includes mandatory financing.
Justification of Budget Request
National Institute of Neurological Disorders and Stroke
Authorizing Legislation: Section 301 and Title IV of the Public Health Service Act, as amended.
Budget Authority (BA):
| FY 2016 Actual |
FY 2017 Annnnualized CR |
FY 2018 President's Budget |
FY 2018 +/- FY 2017 |
|
|---|---|---|---|---|
| BA | $1,692,832,464 | $1,692,915,000 | $1,355,998,000 | -$336,917,000 |
| F T E | 520 | 532 | 532 | 0 |
Program funds are allocated as follows: Competitive Grants/Cooperative Agreements; Contracts; Direct Federal/Intramural and Other.
Director's Overview
Stroke, traumatic brain injury (TBI), chronic pain, epilepsy, dementias, Parkinson’s disease, multiple sclerosis, and other common neurological disorders affect millions of Americans of all ages. Hundreds of rare diseases add to the enormous impact. The National Institute of Neurological Disorders and Stroke (NINDS) supports research to improve the diagnosis, prevention, and treatment of these disorders. Fundamental research on the brain and its diseases is a pivotal aspect of this mission because NIH basic research drives progress against disease throughout the public and private sectors
Clinical trials on prevention and treatment yield the most direct benefit from NINDS research. The cumulative information from many stroke trials, for example, enables physicians to tailor care for each person, which has contributed to a 34 percent decline in age-adjusted stroke death rates in 10 years1. Continuing this trend, in the past year a trial found that pioglitazone, a drug used for type 2 diabetes, prevents recurrent stroke in people with insulin resistance without diabetes, and another trial reported long term outcomes of two surgical procedures, stenting, and endarterectomy, to avoid strokes in people with narrowing of the carotid arteries2. Recent trials for other diseases demonstrated the efficacy of thymectomy, a surgical treatment, for the chronic muscle weakening disease myasthenia gravis, the value of the drug dichlorphenamide for periodic paralysis, and the ineffectiveness of two commonly used drugs for migraine in children and of aggressive blood pressure lowering in persons with acute intracerebral hemorrhage3. In FY 2018, NINDS will continue its longstanding investment in clinical trials for neurological disorders.
Since the Institute’s founding, NINDS has also advanced preclinical therapy development. Successes include the first enzyme therapies for inherited metabolic disorders, several drugs for epilepsy, and pioneering research on devices that connect with the nervous system, including deep brain stimulation technologies and brain-computer interfaces. NINDS translational programs support early stage, exploratory research and more advanced, milestone-driven team projects that bring together academic disease researchers, expert consultants, and contract services to develop drug, cell, gene, and device therapies. These projects bring therapies to clinical testing or “de-risk” the development path to attract private sector investment. In the past year, treatments for lysosomal storage disorders, neuropathy, glioblastoma, stroke, and spinal muscular atrophy have advanced to clinical readiness.
Unfortunately, treatments for many neurological disorders are far from adequate. When progress against a disease is not forthcoming, lack of basic understanding of the underlying biology is usually the major barrier. Time and again, insights from fundamental scientific studies primarily supported by NINDS have launched breakthroughs against disease. In Parkinson’s disease, for example, basic studies on how nerve cells influence one another led to the drug that is the mainstay of current therapy, fundamental investigations of brain circuits that control movement paved the way for deep brain stimulation (DBS) therapy, and gene studies have identified targets for treatments that are now advancing in the development pipeline. Among the many unexpected findings from basic research in 2016, studies on the origins of cells in the early brain revealed how the Zika virus gains access to the developing brain, research on the normal development of the brain’s connections presented clues about autism and on memory impairment, and studies on molecules that underlie our normal ability to sense touch and the body’s position in space led to an explanation for a rare inherited disorder.
In 2014, an NINDS analysis revealed that investigators were submitting a declining number of basic research proposals, which threatened the wellspring of progress. Discussions across NIH and the research community on the state and value of basic research prompted strong reaffirmations of support for basic research by all NIH IC directors4. NINDS reinvigorated its basic research program through improved guidance for peer reviewers, a solicitation with set aside funding for fundamental research, and the Research Program Award (R35), which provides outstanding investigators with freedom and sustained support to undertake ambitious, long-term projects. NINDS will continue to place a high priority on basic research in FY 2018.
Complementing investigator-initiated programs, NINDS targets funding to unmet public health needs and extraordinary scientific opportunities, guided by extensive planning with the scientific community and the public. Among such new and continuing activities for FY 2018:
- For basic research, NINDS is an enthusiastic leader of the Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative, which is developing tools that are revolutionizing basic research on how brain circuits work in health and disease.
- For chronic pain, the Federal Pain Research Strategy, to be completed in 2017, will guide pain research by the NIH Pain Consortium and by other agencies.
- For dementia, the National Alzheimer’s Project Act (NAPA) recognized the impact of not just Alzheimer’s, the most common dementia, but also Alzheimer’s disease-related dementias (ADRDs), which include vascular cognitive impairment/dementia (VCID) and Lewy Body, Parkinson’s, and frontotemporal dementias (FTD).Recommendations from a 2013 NINDS-led ADRD planning process led to several targeted initiatives, and a 2016 meeting updated priorities and timelines to guide activities in FY 2018.
- NINDS is also continuing to support research through initiatives in Parkinson’s disease, muscular dystrophy, stroke, epilepsy, myalgic encephalomyelitis/chronic fatigue syndrome, and other diseases as detailed in the following sections.
Not long ago, neurologists could diagnose diseases of the brain, but do little to treat them. Although medical care is still far from adequate for many neurological disorders, the last two decades have yielded encouraging progress. This includes, for example, more than a dozen drugs for multiple sclerosis, deep brain stimulation therapy for Parkinson’s, and emergency and evidence based preventive interventions for stroke. Previously baffling rare disorders, such as Rett syndrome, spinal muscular atrophy, and muscular dystrophies, whose prospects were especially bleak, are now seeing the first rationally developed therapies entering clinical trials, built on new understanding of the underlying diseases. And astonishing progress is emerging every day from basic research laboratories that bodes well for the future, from nanoscale understanding of how molecules control activity of nerve cells that sense pain and new insights about how dormant genes from viruses may contribute to amyotrophic lateral sclerosis (ALS), to unravelling the mysteries of how the normal brain develops, learns, ignores distractions, and coordinates complex movements. Opportunities for progress against neurological disorders and in promoting optimal brain health throughout life have never been better. NINDS is committed to supporting the highest quality, innovative research by scientists and physicians throughout the United States who can realize this promise.
The multiplicity of neurological disorders, the complexity of the brain, and the difficulty of intervening without disrupting the brain’s indispensable functions present formidable challenges. But there is encouraging progress, with therapies that target the underlying molecular mechanisms of several brain diseases now at various stages of testing. Furthermore, crosscutting scientific progress is increasingly revealing shared causes among diseases and new treatment strategies with potentially widespread uses, and basic science of the brain is poised to make unprecedented progress, driven by –and driving—breathtaking new technologies.
Overall Budget Policy: The FY 2018 President’s Budget request is $1,355.998 million, a decrease of $336.917 million compared to the FY 2017 annualized Continuing Resolution level. These reductions are distributed across all programmatic areas and basic, epidemiology, or clinical research. The Institute continues to hold investigator-initiated research, with rigorous peer review, to be paramount because of its proven track record of driving progress. In times of hypercompetition for funding, NINDS places an especially high priority on supporting early stage investigators and smaller, high quality laboratories, and on maintaining an appropriate balance of basic, translational, and clinical research. Complementing investigator initiated research, the Institute solicits research proposals that target critical unmet public health needs or exceptional scientific opportunities, or provide common resources that enhance productivity of the research community. NINDS emphasizes good stewardship throughout its programs. To inform its decision making, NINDS monitors trends in research portfolios, evaluates specific programs, and engages stakeholders in strategic planning and in priority setting for specific diseases and issues, including health disparities and workforce diversity. By scheduling interim analyses of large clinical trials and by monitoring milestones in large clinical studies and preclinical therapy development projects, NINDS allocates funds to the best current opportunities. Institute policies that strongly favor early stage investigators and an array of training programs for physicians and scientists promote workforce priorities. Finally, NINDS has been among the leaders at NIH and in the scientific community in promoting transparency and reproducibility of research, which is essential to ensure the value of all research investments. To further enhance rigor and reproducibility across all programs, the Institute has appointed a Director of Research Quality.
1 From 2003 to 2013, Circulation 133:447-54 2016
2New England J. Medicine 374:1321-31, 2016; New England J Medicine 374:1021-31, 2016
3New England J Medicine 375:511-22, 2016; Neurology 86:1408-16, 2016; New England J Medicine [online Oct 27 2016]; New England J Medicine 375:1033-43, 2016
4Science 351:1405, 2016 http;//science.sciencemag.org/content/351/6280/1405.1.full
Program Descriptions and Accomplishments:
Channels, Synapses, and Circuits:Ion channels, synapses, and neural circuits are fundamental components of the nervous system. Ion channels control the electrical activity of cells. Synapses are specialized connections by which nerve cells influence the activity of other nerve cells. Circuits formed by networks of interconnected nerve cells carry out the information processing that enables us to perceive, think, and act. In addition to basic research on these fundamental components of the nervous system, the Channel, Synapses and Neural Circuits Program supports studies on dysfunctions of channels, synapses, and circuits that underlie many neurological disorders. Epilepsy, a prime example, is a major focus of the program. Many types of epilepsy affect people of all ages. The causes of epilepsy include gene mutations, head injury, abnormal brain development, and other illnesses. About a third of people with epilepsy do not respond well to any of the available treatments, but there is real promise that new gene discoveries and new neurotechnologies to monitor neural circuit activity may lead to better treatments for those with medically refractory epilepsy. There are now several examples in which identification of rare mutations that cause a particular child’s epilepsy have led directly to the improved treatment for particular children, although treating all of the many types of epilepsy will present formidable challenges. A major priority for NINDS is to understand how epilepsy first develops with an eye toward informing the development of preventative treatments. Since 2000, the NINDS Epilepsy Benchmarks for Epilepsy Research have brought together disease advocates, the scientific community, and NIH to work toward the shared goal of curing all forms of epilepsy.
Infrastructure, Training programs, and Resources:The Division of Clinical Research (DCR) supports infrastructure and programs for clinical research, including early and advanced phase clinical trials and large epidemiological studies. DCR works closely with disease experts across all NINDS extramural programs. DCR has significantly increased the efficiency and effectiveness of large scale clinical trials by enforcing milestones for progress, providing resources to improve patient access and recruitment, and developing multi-site clinical networks. DCR pioneered innovations for multi-site trials, such as use of a single Institutional Review Board, that reduce the time and cost required to launch new trials, and has for several years directed all clinical trials applications through specific Notices of Funding Opportunities to ensure optimal peer review. These practices are now adopted NIH-wide. The DCR Common Data Elements (CDE) Program works with the research and patient advocacy communities to develop standards for specific disorders that facilitate comparison and sharing of clinical data across studies, enhancing the value of these major clinical investments.
The Office of Training, Career Development, and Workforce Diversity coordinates NINDS extramural programs for training and career development tailored to the specific needs of physicians and scientists at different stages of their careers. Following the advice of planning panels on workforce diversity, the Institute integrated its programs on diversity within this office. The Office of International Activities leads international collaborations that advance the NINDS mission and coordinates the Institute’s global health activities, working closely with the Fogarty International Center.
A full range of NINDS programs in training and career development are also continuing, including individual and institutional grants at the graduate, post-doctoral, and career development levels, and an intensive training course in clinical trials methods for fellows and faculty in the clinical neurosciences. Many of the programs target special needs, such as promoting diversity and enabling neurosurgeons to accommodate research preparation into their demanding training requirements. NINDS has redesigned its early stage training programs to improve opportunities for early-career neuroscientists and bolster their transition to independent research careers. These changes include a newly designed F32 postdoctoral fellowship program for early stage post-doctoral students, a new postdoctoral K01 program that enables advanced post-doctoral students to develop an independent research program, and a new NIH Blueprint predoctoral/postdoctoral (F99/K00) award to help recruit and retain faculty who represent the diverse make-up of our country. NINDS is also developing a new Mentor Award that recognizes the value of outstanding mentorship in developing each new generation of exceptional neuroscientists.
Neural Environment:The Neural Environment Program supports basic research on the neural environment, on diseases in which its disruption plays a major role, and on translation of basic scientific knowledge into diagnostic tools, preventive measures, and targeted therapies. Non-nerve cells in the brain, called glial cells, are as numerous as nerve cells in the brain. Glial cells, together with specialized blood vessels and immune cells in the brain, maintain the local environment around nerve cells, fighting infections, and controlling which molecules enter brain tissue from the circulating blood. Recent research has focused increasing attention on these cells, with demonstrations that they actively shape brain development and synapse function and may provide clues to a host of diseases, including Alzheimer’s, autism, and recovery from brain and spinal cord injury. Neurological disorders can result when non-neuronal cells are compromised; when these cells become aggressors in inflammatory or autoimmune disorders; when cells form tumors; when viruses, bacteria, or parasites infect the nervous system; or when the blood supply to brain cells is compromised, as in stroke. The Program coordinates NINDS’ stroke activities, which span several parts of the Institute and have had a major impact on public health. Multiple sclerosis is another common disorder that affects glial cells. Basic NIH supported research on immunology of the brain and on glial cells has fostered private sector development of more than a dozen drugs for multiple sclerosis over the last two decades, and hospitalization rates due to multiple sclerosis have declined by 75 percent over that period5. Progress against brain tumors, another focus of this program, has been much more difficult to achieve, but a wide range of new studies are revealing potential strategies. For all diseases within the NINDS mission, the Institute coordinates closely with other components of NIH when our missions intersect; NINDS collaboration with NHLBI on stroke, NCI on cancer, and NIAID on multiple sclerosis and infectious diseases of the nervous system are among the many examples.
5Neurology 84:929-37 2014
Neurodegeneration:The Neurodegeneration Program focuses on adult onset neurodegenerative diseases, in which brain cells progressively die. The National Alzheimer’s Disease Project Act (NAPA) recognized the public health importance of not just Alzheimer’s disease, but also other related dementias that arise from neurodegeneration. Vascular cognitive impairment/dementia (VCID) is the second most common dementia. Frontotemporal dementia (FTD) is the most common dementia in people under 60 years old, and Lewy body dementia is also an important type. NINDS works closely with NIA on neurodegenerative diseases of aging. NIA leads research on Alzheimer’s disease, and NINDS plays a leading role in research against Alzheimer’s disease related dementias and on Parkinson’s disease, Huntington’s disease, and amyotrophic lateral sclerosis (ALS, or Lou Gehrig’s disease). Research has increasingly revealed connections among all of these neurodegenerative disorders. VCID is so closely intertwined with Alzheimer’s that most people have a combination of the two. Research has found strong connections between stroke risk factors and dementias, including Alzheimer’s, at every level of investigation from molecular mechanisms to pathology, and progress against stroke may also be having an impact on the prevalence of Alzheimer’s disease6. With regard to underlying disease mechanisms, a major unifying theme is the recognition that in many neurodegenerative disorders certain proteins form abnormal aggregates. Research on genetic contributions to neurodegenerative diseases has also driven progress and revealed unsuspected connections, as that between ALS and FTD, as well as between inherited and non-inherited disease, as in Parkinson’s. Several drugs targeting steps in the underlying diseases revealed through genetic and molecular studies are now at various stages of testing.
6New England Journal of Medicine 374:523-32 2016
Neurogenetics:Gene defects cause hundreds of rare diseases that affect the nervous system, including ataxias, Down syndrome, dystonias, fragile X syndrome, lysosomal storage diseases, muscular dystrophies, peripheral neuropathies, Rett syndrome, spinal muscular atrophy, Tourette syndrome, and tuberous sclerosis, among many others. Genes also influence susceptibility to common neurological disorders. The Neurogenetics Program supports research on genes that cause or influence neurological disorders, molecular mechanisms by which these genes act, genetic animal and cell models of human disease, and development of treatments for neurogenetic disorders, from the laboratory to the clinic. Basic research to understand how genes and the environment orchestrate brain development is also a key aspect of the Neurogenetics Program. Over the last two decades, research has identified hundreds of genes related to neurological disorders, which has led to tests that enable physicians to diagnose a disease months or even years more quickly than before, reducing the “diagnostic odyssey” that families who have a child with a rare disease often confront.Gene findings have also led to animal models that provide insights about the underlying mechanisms of human diseases and allow testing of strategies for therapy development that target these disease mechanisms. In late 2016, progress in understanding spinal muscular atrophy led to the the approval of the first FDA approved therapy targeting the mechanism of this disorder, and therapies for several other rare genetic diseases area at various stages of development in the public and private sector.
Repair and Plasticity:The Repair and Plasticity Program leads NINDS research on traumatic brain injury (TBI), spinal cord injury, and peripheral nerve injury. Research covers the full spectrum, including mechanisms of immediate damage, delayed effects in the hours after initial injury, laboratory development of interventions that minimize damage or stimulate nervous system repair in animal models, and clinical testing in people. The Program also supports fundamental studies of neural plasticity, that is, the ability of the brain and nervous system to change, which has broad implications for recovery following injury or disease. This includes studies of stem cells in the brain. Research in FY 2016, for example, showed that the continued generation of new nerve cells in adult brains contributes to learning and memory and that intense rehabilitation training can drive significant changes in the structure of brain cells adjacent to an injury. The Repair and Plasticity Program also leads the longstanding NINDS Neural Interfaces Program, which pioneered technology for devices that connect to the nervous system to compensate for lost functions, including therapeutic brain stimulation technologies and brain computer interfaces that enable paralyzed people to control devices with their thoughts.
Systems and Cognitive Neuroscience:Systems of interconnected nerve circuits in the brain, spinal cord, and body control learning, memory, attention, language, thinking, emotion, sensation, movement, and response to pain, as well as sleep, feeding, and drinking. The Systems and Cognitive Neuroscience Program supports research on how the brain carries out these complex functions, on their disruption in neurological disorders, and on promoting recovery. Non-invasive brain imaging is an important research tool for this program, including monitoring of brain activity associated with specific types of thinking and behavior. NINDS is a partner in the Human Connectome Project, which has provided unparalleled data for neuroscientists on how parts of the brain interact with one another. Scientists are now building on the foundation of normal brain connectomes to examine how connectivity is altered during disease. Chronic pain disorders, including migraine and other headaches, are among the most prevalent of all medical conditions and are a high priority for this Program. NINDS leads NIH pain research, which is coordinated through the Office of Pain Policy and the NIH Pain Consortium. The NINDS Office of Pain Policy manages the Interagency Pain Research Portfolio, a database that provides the public and the research community with information on the breadth and details of pain research and training activities supported by six Federal agencies. Research to expedite the development of more effective non-opioid pain agents is a high priority for NINDS because of the public health impact of opioid addiction.
Translational Research:The Division of Translational Research (DTR) leads NINDS preclinical therapy development activities for all diseases within the Institute’s mission. DTR supports the development of drugs, devices, and biologics, including cell and gene therapies, either toward further clinical development by NIH or by “de-risking” the path to treatment to the point where the private sector is willing to invest in further development. DTR preclinical therapy development experts work closely with disease-specific experts in extramural programs across the Institute and with the Division of Clinical Research to advance these interventions to first-in-human studies. The Innovation Grants to Nurture Initial Translational Efforts (IGNITE) Program provides a suite of funding opportunities for early stages of therapy development, for example, validation of assays to evaluate candidate drugs, demonstrations that proposed therapies have sufficient biological activity to warrant further development, and development of model systems for therapy development. Successful projects from IGNITE (or other promising research) progress into the Cooperative Research to Enable and Advance Translational Enterprises (CREATE) programs for biologics (such as large biological molecules, gene therapies, and cell therapies) and for devices that advance therapies into early phase clinical trials. The Blueprint Neurotherapeutics Network supports the development of small molecule drugs. These advanced development programs gate funding based on rigorous milestones of progress and provide expert consultation and contract services that are often not readily available to academic investigators. DTR also coordinates all NINDS small business programs, which support innovative ideas at different stages of development, including applied bench research, translational research, and early stage clinical trials.
Intramural Research Program (IRP):IRP conducts research on the NIH campus in Bethesda, Maryland. The Program spans basic and translational neuroscience, neurology, and neurosurgery, and maintains its historical strengths in neuromuscular diseases, neurogenetics, neuroimmunology, membrane biochemistry and biophysics, and neurophysiology at the systems and synaptic level. The Mark O. Hatfield Clinical Center on the NIH campus in Bethesda, which is a hospital totally dedicated to clinical research, and the Porter Neuroscience Research Center, which integrates neuroscience across NIH ICs and disciplinary boundaries, provide unique resources to the Intramural Research Program. Ongoing basic research ranges from studies of individual molecules that control nerve cell behavior to advancing human brain imaging. Early phase clinical studies continue to be a strong aspect of the IRP, with studies now underway at the Clinical center on Parkinson’s, epilepsy, multiple sclerosis, dystonia, neuropathies, and several other diseases, using drugs, invasive and noninvasive brain stimulation techniques, and gene therapy.
Research Management and Support (RMS):RMS activities provide administrative, budgetary, logistical, and scientific support in the review, award, and monitoring of research grants, training awards, and research and development contracts. RMS functions also encompass strategic planning, coordination, and evaluation of the Institute’s programs, regulatory compliance, international coordination, and liaison with other Federal agencies, Congress, and the public.
Details of Full-Time Equivalent Employment (FTE's)
| OFFICE/DIVISION | FY 2016 Final |
FY 2017 Annualized CR |
FY 2018 President's Budget |
||||||
|---|---|---|---|---|---|---|---|---|---|
| Civilian | Military | Total | Civilian | Military | Total | Civilian | Military | Total | |
| Office of the Director | |||||||||
| Direct: | 6 | - | 6 | 8 | - | 8 | - | - | 8 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 6 | - | 6 | 8 | - | 8 | - | - | 8 |
| Office of Management | |||||||||
| Direct: | 33 | - | 33 | 34 | - | 34 | 34 | - | 34 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 33 | - | 33 | 34 | - | 34 | 34 | - | 34 |
| Office of Communications and Public Liaison | |||||||||
| Direct: | 10 | - | 10 | 10 | - | 10 | 10 | - | 10 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 10 | - | 10 | 10 | - | 10 | 10 | - | 10 |
| Office of Science Policy and Planning | |||||||||
| Direct: | 5 | - | 5 | 5 | - | 5 | 5 | - | 5 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 5 | - | 5 | 5 | - | 5 | 5 | - | 5 |
| Office of Scientific Liaison | |||||||||
| Direct: | 3 | - | 3 | 3 | - | 3 | 3 | - | 3 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 3 | - | 3 | 3 | - | 3 | 3 | - | 3 |
| Division of Clinical Research | |||||||||
| Direct: | 13 | - | 13 | 15 | - | 15 | 15 | - | 15 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 13 | - | 13 | 15 | - | 15 | 15 | - | 15 |
| Division of Translational Research | |||||||||
| Direct: | 18 | - | 18 | 18 | - | 18 | 18 | - | 18 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 18 | - | 18 | 18 | - | 18 | 18 | - | 18 |
| Division of Intramural Research | |||||||||
| Direct: | 311 | 9 | 320 | 315 | 8 | 323 | 312 | 8 | 320 |
| Reimbursable: | 6 | - | 6 | 10 | - | 10 | 13 | - | 13 |
| Total: | 317 | 9 | 326 | 325 | 8 | 333 | 325 | 8 | 333 |
| Division of Extramural Research | |||||||||
| Direct: | 56 | - | 56 | 56 | - | 56 | 56 | - | 56 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 56 | - | 56 | 56 | - | 56 | 56 | - | 56 |
| Division of Neuroscience | |||||||||
| Direct: | 50 | - | 50 | 50 | - | 50 | 50 | - | 50 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 50 | - | 50 | 50 | - | 50 | 50 | - | 50 |
| Total | 511 | 9 | 520 | 524 | 8 | 532 | 524 | 8 | 532 |
Includes FTEs whose payroll obligations are supported by the NIH Common Fund.
FTEs supported by funds from Cooperative Research and Development Agreements.
| FISCAL YEAR | Average GS Grade |
|---|---|
| 2014 | 12.0 |
| 2015 | 12.0 |
| 2016 | 12.3 |
| 2017 | 12.3 |
| 2018 | 12.3 |
Detail of Positions3
(Dollars in Thousands)
| GRADE | FY 2016 Final Actual |
FY 2017 Annualized CR |
FY 2017 President's Budget |
|---|---|---|---|
| Total, ES Positions | 1 | 1 | 1 |
| Total, ES Salary | 185,100 | 185,100 | 185,100 |
| GM/GS-15 | 46 | 47 | 47 |
| GM/GS-14 | 50 | 50 | 50 |
| GM/GS-13 | 89 | 92 | 92 |
| GS-12 | 60 | 62 | 62 |
| GS-11 | 31 | 31 | 31 |
| GS-10 | 3 | 3 | 3 |
| GS-9 | 17 | 20 | 20 |
| GS-8 | 15 | 15 | 15 |
| GS-7 | 10 | 10 | 10 |
| GS-6 | 1 | 1 | 1 |
| GS-5 | 0 | 0 | 0 |
| GS-4 | 0 | 0 | 0 |
| GS-3 | 2 | 2 | 2 |
| GS-2 | 0 | 0 | 0 |
| GS-1 | 0 | 0 | 0 |
| Subtotal | 324 | 333 | 333 |
| Grades established by Act of July 1, 1944 (42 U.S.C. 207): | 0 | 0 | 0 |
| Assistant Surgeon General | 0 | 0 | 0 |
| Director Grade | 3 | 3 | 3 |
| Senior Grade | 1 | 1 | 1 |
| Full Grade | 2 | 2 | 2 |
| Senior Assistant Grade | 2 | 2 | 2 |
| Assistant Grade | 0 | 0 | 0 |
| Subtotal | 8 | 8 | 8 |
| Ungraded | 0 | 0 | 0 |
| Total permanent positions | 337 | 346 | 346 |
| Total positions, end of year | 546 | 559 | 559 |
| Total full-time equivalent (F T E) employment, end of year | 520 | 532 | 532 |
| Average ES salary | 185,100 | 185,100 | 185,100 |
| Average GM/GS grade | 12.3 | 12.3 | 12.3 |
| Average GM/GS salary | 105,124 | 107,227 | 107,227 |
3 Includes FTEs whose payroll obligations are supported by the NIH Common Fund.
