- Organization Chart
- Appropriation Language
- Amounts Available for Obligation
- Budget Mechanism Table
- Major Changes in Budget Request
- Summary of Changes
- Budget Graphs
- Budget Authority by Activity
- Authorizing Legislation
- Appropriations History
- Justification of Budget Request
- Budget Authority by Object Class
- Salaries and Expenses
- Detail of Full-Time Equivalent Employment (FTE)
- Detail of Positions
Organization Chart
National Institutes of Health
National Institute of Neurological Disorders and Stroke
For carrying out section 301 and title IV of the PHS Act with respect to neurological disorders and stroke, [$1,696,139,000] $1,659,416,000.
Amounts Available for Obligation 1
(Dollars in Thousands)
| Source of Funding | F Y 2015 Actual |
F Y 2016 Enacted | F Y 2016 President's Budget |
|---|---|---|---|
| Appropriation | $1,605,205 | $1,696,139 | $1,695,180 |
| Mandatory Appropriation: (non-add) | |||
| Type 1 Diabetes | (0) | (0) | (0) |
| Other Mandatory financing | 0 | (0) | (37,764) |
| Rescission | 0 | 0 | 0 |
| Sequestration | 0 | 0 | 0 |
| FY 2015 First Secretary's Transfer | 0 | 0 | 0 |
| FY 2015 Second Secretary's Transfer | 0 | 0 | 0 |
| Subtotal, adjusted appropriation OAR HIV/AIDS Transfers National Children's Study Transfers |
$1,605,205 -598 0 |
$1,696,139 -959 0 |
$1,695,180 0 0 |
| Subtotal, adjusted budget authority Unobligated balance, start of year Unobligated balance, end of year |
$1,604,607 0 0 |
$1,695,180 0 0 |
$1,695,180 0 0 |
| Subtotal, adjusted budget authority Unobligated balance lapsing |
$1,604,607 -26 |
$1,695,180 0 |
$1,695,180 0 |
| Total obligations | $1,604,581 | $1,6095,180 | $1,695,180 |
1 Excludes the following amounts for reimbursable activities carried out by this account:
FY 2015 - $15,430 FY 2016 - $15,456 FY 2017 - $15,488
Budget Mechanism - Total 1
(Dollars in Thousands)
| MECHANISM | FY 2015 Actual |
FY 2016 Enacted | FY 2017 President's Budget |
FY 20173 +/- FY 2016 |
||||
|---|---|---|---|---|---|---|---|---|
| No. | Amount | No. | Amount | No. | Amount | No. | Amount | |
| Research Projects: | ||||||||
| Noncompeting | 1,884 | $769,056 | 1,944 | $834,376 | 2,001 | $852,382 | 57 | $18,006 |
| Administrative Supplements | (92) | 5,240 | (95) | 5,000 | (95) | 5,000 | (0) | 0 |
| Competing: | ||||||||
| Renewal | 137 | 65,576 | 137 | 65,576 | 124 | 47,135 | -13 | -18,441 |
| New | 682 | 245,292 | 682 | 245,525 | 618 | 234,641 | -64 | -10,884 |
| Supplements | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Subtotal, Competing | 819 | $310,869 | 819 | $311,101 | 742 | $281,776 | -77 | $29,325 |
| Subtotal, RPGs | 2,708 | $1,085,165 | 2,763 | $1,150,477 | 2,743 | $1,139,158 | -20 | $11,319 |
| SBIR/STTR | 94 | 45,221 | 95 | 50,560 | 103 | 54,834 | 8 | 4,274 |
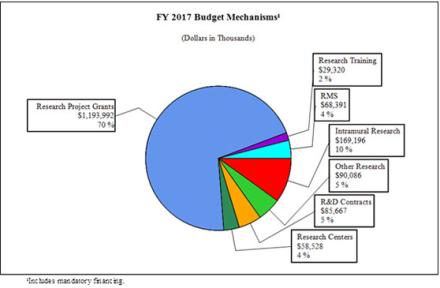
| Research Project Grants | 2,797 | $1,130,386 | 2,858 | $1,1201,037 | 2,846 | $1,193,992 | -12 | -$7,045 |
Research Centers: |
||||||||
| Specialized/Comprehensive | 53 | $54,193 | 61 | $58,528 | 61 | $58,528 | 0 | 0 |
| Clinical Research | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Biotechnology | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Comparative Medicine | 0 | 588 | 0 | 600 | 0 | 0 | 0 | 0 |
| Research Centers in Minority Institutions | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Research Centers | 53 | $54,781 | 61 | $58,528 | 61 | $58,528 | 0 | 0 |
Other Research: |
||||||||
| Research Careers | 205 | $34,704 | 199 | $35,398 | 199 | $345,398 | 0 | $0 |
| Cancer Education | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Cooperative Clinical Research | 67 | 17,635 | 69 | 21,017 | 70 | 21,017 | 1 | 208 |
| Biomedical Research Support | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Minority Biomedical Research Support | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Other | 132 | 30,541 | 139 | 33,671 | 146 | 33,671 | 7 | 1,603 |
| Other Research | 404 | $82,880 | 407 | $90,086 | 415 | $90,086 | 8 | $1,811 |
| Total Research Grants | 3,254 | $1,268,047 | 3,326 | $1,342,606 | 3,322 | $1,342,606 | -4 | -$5,234 |
Ruth L. Kirschstein Training Awards: |
FTTPs |
FTTPs |
FTTPs |
|||||
| Individual Awards | 340 | 14,176 | 340 | $14,460 | 340 | $14,749 | 0 | $289 |
| Institutional Awards | 299 | 14,005 | 299 | 14,285 | 299 | 14,571 | 0 | 286 |
| Total Research Training | 639 | $28,182 | 639 | $28,745 | 639 | $29,320 | 0 | $575 |
Research & Development Contracts |
57 |
$84,987 |
100 |
$85,667 |
100 |
$85,667 |
0 |
$0 |
| SBIR/STTR (non-add)2 | (2) | (383) | (2) | (850) | (2) | (850) | (0) | (0) |
| Intramural Research | 325 | 159,567 | 325 | $165,878 | 325 | 169,196 | 0 | 3,318 |
| Research Management and Support | 192 | 63,824 | 197 | 67,050 | 197 | 68,391 | 0 | 1,341 |
| Res. Management & Support (SBIR Admin) (non-add) | (0) | (463) | (0) | (500) | (0) | (500) | (0) | (0) |
| Office of the Director - Appropriation2 | 325 | 159,567 | 325 | $165,878 | 325 | 169,196 | 0 | 3,318 |
| Office of the Director - Other | 192 | 63,824 | 197 | 67,050 | 197 | 68,391 | 0 | 1,341 |
| ORIP/SEPA (non-add)2 | (0) | (463) | (0) | (500) | (0) | (500) | (0) | (0) |
| Common Fund (non-add)2 | (0) | (463) | (0) | (500) | (0) | (500) | (0) | (0) |
| Buildings and Facilities | 0 | 0 | 0 | 0 | ||||
| Appropriation | 0 | 0 | 0 | 0 | ||||
| Type 1 Diabetes | 0 | 0 | 0 | 0 | ||||
| Program Evaluation Financing | 0 | 0 | 0 | 0 | ||||
| Cancer Initiative Mandatory Financing | 0 | 0 | 0 | 0 | ||||
| Other Mandatory Financing | 0 | 0 | -35,764 | -35,764 | ||||
| Subtotal, Labor/HHS Budget Authority | 0 | $1,604,607 | 0 | $1,695,180 | 0 | $1,659,416 | 0 | -$35,764 |
| Interior Appropriation for Superfund Res. | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total, NIH Discertionary B.A. | 0 | $1,604,607 | 0 | $1,695,180 | 0 | $1,659,416 | 0 | -$35,764 |
| Type 1 Diabetes | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Proposed Law Funding | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Cancer Initiative Mandatory Funding | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Other Mandatory Financing | 0 | 0 | 0 | 0 | 0 | 35,764 | 0 | 35,764 |
| Total, NIH Budget Authority | 0 | $1,604,607 | 0 | $1,695,180 | 0 | $1,695,180 | 0 | 0 |
| Program Evaluation Financing | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total Program Level | 0 | $1,604,607 | 0 | $1,695,180 | 0 | $1,695,180 | 0 | 0 |
1 All Subtotal and Total numbers may not add due to no rounding.
2 All numbers in italics and brackets are non-add.
3 Includes mandatory financing.
Major Changes in the Fiscal Year 2017 President's Budget Request
Small Business Innovation Research/ Small Business Technology Transfer SBIR/STTR (+$4.274 million, total $54.834 million):
NINDS will increase resources to support SBIR and STTR projects to reflect a 3.65 percent share of NINDS’s budget.
Other Research (+$1.811 million, total $90.086 million):
NINDS will increase resources in Other Research to support Resource-Related Research Projects (R24) for the BRAIN Initiative.
Research Training (+$0.575 million, total $29.320 million):
NIH will provide an increase of two percent over FY 2016 for stipend levels for pre-doctoral and post-doctoral trainees. The requested increase will help sustain the development of a highly qualified biomedical research workforce.
Summary of Changes 1
(Dollars in Thousands)
| F Y 2016 Enacted | $1,695,180 |
|---|---|
| F Y 2017 President's Budget | $1,695,180 |
| Net change | $0 |
| 2017 President's Budget |
Change from F Y 2017 |
|||
|---|---|---|---|---|
| CHANGES | FTE's | Budget Authority |
FTE's | Budget Authority |
| A. Built-in: 1. Intramural research: |
||||
| a. Annualization of January 2016 pay increase & benefits |
$53,855 | $133 | ||
| b. January FY 2017 pay increase & benefits | 53,855 | 400 | ||
| c. Two less days of pay | 53,855 | -407 | ||
| d. Differences attributable to change in FTE | 53,379 | 0 | ||
| e. Payment for centrally furnished services | 28,908 | 3,171 | ||
| f. Increased cost of laboratory supplies, materials, other expenses, and non-recurring costs |
86,434 | 181 | ||
| Subtotal | $3,479 | |||
2. Research Management and Support: |
||||
| a. Annualization of January 2016 pay increase & benefits |
$29,634 | $72 | ||
| b. January FY 2017 pay increase & benefits |
29,634 | 217 | ||
| c. Two less days of pay | 29,634 | -221 | ||
| d. Differences attributable to change in FTE | 29,634 | 0 | ||
| e. Payment for centrally furnished services | 7,748 | 1,029 | ||
| f. Increased cost of laboratory supplies, materials, other expenses, and non-recurring costs |
31,009 | 83 | ||
| Subtotal | $1,180 | |||
| Subtotal, Built-in | $4,659 | |||
| 2017 President's Budget |
Change from F Y 2016 |
|||
|---|---|---|---|---|
| CHANGES | No. | Amount | No. | Amount |
| B. Program: | ||||
| 1. Research Project Grants: | ||||
| a. Noncompeting | 2,001 | $857,382 | 57 | $18,006 |
| b. Competing | 742 | 281,776 | -77 | -29,325 |
| c. SBIR/STTR | 103 | 54,834 | 8 | 4,274 |
| Subtotal, RPGs | 2,846 | $1,193,992 | -12 | -$7,045 |
| 2. Research Centers | 61 | $58,528 | 0 | 0 |
| 3. Other Research | 415 | 90,086 | 8 | 1,811 |
| 4. Research Training | 639 | 29,320 | 0 | 575 |
| 5. Research and Development Contracts | 100 | 85,667 | 0 | 0 |
| Subtotal, Extramural | $1,457,593 | -$4,659 | ||
| FTE's | FTE's | |||
| 6. Intramural Research | 325 | $169,196 | 0 | -$161 |
| 7. Research Management and Support | 197 | 68,391 | 0 | 161 |
| 8. Construction | 0 | 0 | ||
| 9. Buildings and Facilities | 0 | 0 | ||
| Subtotal, Program | 522 | $1,695,180 | 0 | -$4,659 |
| Total Changes | 0 | |||
1Includes mandatory financing.
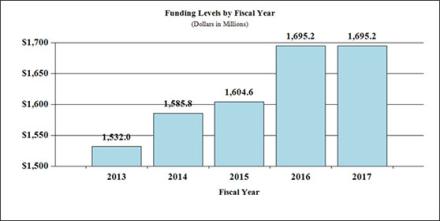
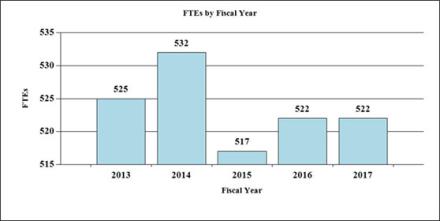
Fiscal Year 2017 Budget Graphs
History of Budget Authority and FTE's:
Distribution by Mechanism:
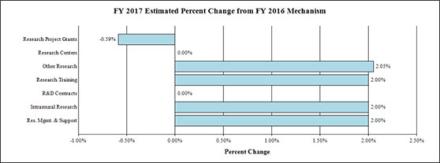
Change by Selected Mechanisms:
Budget Authority by Activity
(Dollars in Thousands)
| FY 2015 Actual |
FY 2016 Enacted |
FY 2017 President's Budget2 |
FY 2017 +/- FY 2016 |
|||||
|---|---|---|---|---|---|---|---|---|
| Extramural Research Detail: |
FTEs | Amount | FTEs | Amount | FTEs | Amount | FTEs | Amount |
| Channels, Synapses & Circuits | 194,291 | 205,689 | 205,034 | -$655 | ||||
| Infrastructure, Training Programs, and Resources | 132,059 | 139,807 | 139,361 | -445 | ||||
| Neural Environment | 229,097 | 242,537 | 241,765 | -773 | ||||
| Neurodegeneration | 217,254 | 229,999 | 229,267 | -733 | ||||
| Neurogenetics | 228,184 | 241,572 | 240,802 | -770 | ||||
| Repair & Plasticity | 135,940 | 142,857 | 142,402 | -455 | ||||
| Systems & Cognitive Neuroscience | 191,064 | 202,274 | 201,629 | -644 | ||||
| Translational Research | 54,327 | 57,517 | 57,334 | -183 | ||||
| Subtotal, Extramural | $1,381,216 | $1,462,252 | $1,457,593 | -$4,659 | ||||
Intramural Research |
325 | $159,567 | 325 | $165,878 | 325 | $169,196 | 0 | $3,318 |
| Research Management & Support | 192 | $63,824 | 197 | $67,050 | 197 | $68,391 | 0 | $1,341 |
| TOTAL | 517 | $1,604,607 | 522 | $1,695,180 | 522 | $1,695,180 | 0 | $0 |
1 Includes F T Es whose payroll obligations are supported by the NIH Common Fund.
2 Includes mandatory financing.
Authorizing Legislation
| PHS Act/ Other Citation |
U.S. Code Citation |
2016 Amount Authorized |
F Y 2016 Enacted |
2017 Amount Authorized |
F Y 2017 PB1 |
|
|---|---|---|---|---|---|---|
| Research and Investigation | Section 301 | 42§241 | Indefinite | $1,695,180,000 | Indefinite | $1,659,416,000 |
| National Institute of Neurological Disorders and Stroke |
Section 401(a) | 42§281 | Indefinite | Indefinite | ||
| Total, Budget Authority | $1,695,180,000 | $1,659,416,000 |
1 Includes mandatory financing.
Appropriations History
| Fiscal Year | Budget Estimate to Congress | House Allowance | Senate Allowance | Appropriation |
|---|---|---|---|---|
| 2007 | $1,524,750,000 | $1,524,750,000 | $1,537,703,000 | $1,534,757,000 |
| Rescission | $0 | |||
| 2008 | $1,537,019,000 | $1,559,106,000 | $1,573,268,000 | $1,571,353,000 |
| Rescission | $27,452,000 | |||
| 2009 | $1,545,397,000 | $1,598,521,000 | $1,588,405,000 | $1,593,344,000 |
| Rescission | $0 | |||
| Supplemental | $8,212,000 | |||
| 2010 | $1,612,745,000 | $1,650,253,000 | $1,620,494,000 | $1,636,371,000 |
| Rescission | $0 | |||
| 2011 | $1,681,333,000 | $1,678,696,000 | $1,636,371,000 | |
| Rescission | $14,368,312 | |||
| 2012 | $1,664,253,000 | $1,664,253,000 | $1,603,741,000 | $1,629,445,000 |
| Rescission | $3,079,651 | |||
| 2013 | $1,624,707,000 | $1,629,631,000 | $1,626,365,349 | |
| Rescission | $3,252,731 | |||
| Sequestration | $81,632,357 | |||
| 2014 | $1,642,619,000 | $1,631,703,000 | $1,587,982,000 | |
| Rescission | $0 | |||
| 2015 | $1,608,461,000 | $1,605,205,000 | ||
| Rescission | $0 | |||
| 2016 | $1,660,375,000 | $1,656,758,000 | $1,694,758,000 | $1,696,139,000 |
| Rescission | $0 | |||
| 20171 | $1,695,180,000 |
1 Includes mandatory financing.
Justification of Budget Request
National Institute of Neurological Disorders and Stroke
Authorizing Legislation: Section 301 and Title IV of the Public Health Service Act, as amended.
Budget Authority (BA):
| FY 2015 Actual |
FY 2016 Enacted | FY 2017 President's Budget |
FY 2017 +/- FY 2016 |
|
|---|---|---|---|---|
| BA | $1,604,607,000 | $1,695,180,000 | $1,695,180,000 | $0 |
| F T E | 517 | 522 | 522 | 0 |
Program funds are allocated as follows: Competitive Grants/Cooperative Agreements; Contracts; Direct Federal/Intramural and Other.
Director's Overview
The National Institute of Neurological Disorders and Stroke (NINDS) supports research to better understand the brain and nervous system and to improve the diagnosis, treatment, and prevention of nervous system disorders. Stroke, traumatic brain injury (TBI), dementia, chronic pain, epilepsy, Parkinson’s disease, multiple sclerosis, and other all too familiar disorders of the nervous system affect millions of American families. Hundreds of rare and devastating neurological diseases add to the enormous public health burden.
To maximize its impact, NINDS monitors trends in research portfolios, evaluates specific programs, and engages stakeholders in strategic planning and planning for specific diseases and priorities, including heath disparities and workforce diversity. By scheduling interim analyses of large clinical trials and monitoring milestones in preclinical therapy development projects, NINDS shifts funds to the best current opportunities. Institute policies that strongly favor early stage investigators and an array of training programs for physicians and scientists promote workforce priorities. Common research resources enhance sharing and productivity. Although NINDS solicits grant proposals to address unmet public health needs or scientific opportunities, NINDS continues to hold investigator-initiated research paramount because of its proven record of driving breakthroughs. Finally, NINDS has been among the leaders of the scientific community in promoting transparency and reproducibility of research.
Opportunities arising from advances in neuroscience have naturally led many basic scientists to shift toward more applied research. However, NINDS identified a multi-year decline in grant applications in basic research that might endanger future progress against disease in both the public and private sector. Several NINDS actions have stopped this downward trend, including a solicitation for fundamental neuroscience research that will continue in FY 2017. A new NINDS Research Program Award, described below, emphasizes the long-term significance of research and will be especially valuable for basic investigators. Reinforcing the emphasis on basic research, NINDS is an enthusiastic partner in the Brain Research through Advancing Innovative Neurotechnologies (BRAIN) initiative, which is developing technologies to study how brain circuits enable us to perceive, think, and act. In FY 2015, for example, investigators labeled and erased specific memory traces in animals’ brains using optogenetics, which makes nerve cells’ activity subject to control via light pulses, and wirelessly controlled the path a mouse walks using an “optofluidic” brain probe a tenth the width of a human hair that can deliver light or minute doses of a drug. The torrent of complex data from the Initiative will spur innovation in informatics and computational science that is likely to spillover far beyond neuroscience.
The National Alzheimer’s Project Act recognized the public health imperative of not just Alzheimer’s disease, the most common dementia, but also of Alzheimer’s disease related dementias (ADRDs). Vascular cognitive impairment, in which brain blood vessels are affected, is so intertwined with Alzheimer’s disease that most dementia is a combination of the two. Frontotemporal dementia (FTD) is the most common dementia in people under age 60. Lewy body and Parkinson’s dementias also contribute to the immense burden. The National Plan to Address Alzheimer’s Disease has incorporated recommendations from a 2013 NINDS-led summit on ADRDs, which, together with advice from the follow up ADRD Summit 2016, which NINDS is leading, will continue to guide NIH programs to address all dementias.
The U.S. age-adjusted stroke death rate declined by 77 percent from 1969 to 2013.1 Promoting brain health to prevent strokes may also be bending the curve on the incidence of dementia.2 Many NIH clinical studies contributed to progress against stroke by providing evidence to tailor preventive care for individual patients. Continuing this trend, in 2015 the SPRINT clinical trial, which NHLBI led with NINDS support, demonstrated that, for people with certain common risk factors, more stringent than usual blood pressure control lowered the risk of death from heart attack or stroke by almost a quarter compared to less aggressive care. This is a major game changer. Also in 2015, decades of public and private research led to the most significant advance in emergency care for stroke since NINDS developed treatment with the clot busting drug tPA in the 1990s. Clinical trials in several countries found that devices that directly remove an offending clot from a blocked artery can provide striking clinical benefit when tPA does not restore blood flow to the brain in severe strokes. An NINDS clinical trial is now investigating how to optimize patient selection to maximize the benefit.
NINDS and NHLBI are jointly developing the Strategies to Innovate EmeRgeENcy Care Clinical Trials (SIREN) program, which, beginning in FY 2017, will enhance emergency clinical research for both Institute’s missions. SIREN complements StrokeNet, a multi-site clinical network for clinical trials in prevention, treatment, and rehabilitation, which arose from an intensive stroke priority setting process. Other ongoing NINDS initiatives also respond to both strategic planning and specific disease priorities. For example, the Epilepsy Centers without Walls address priorities from the Epilepsy Benchmarks, including a focus on disease modifying and preventive rather than symptomatic therapies. The Anticonvulsant Screening Program, which has contributed to several epilepsy drugs now on the market, is similarly shifting emphasis in response to a 2015 evaluation. Resource programs, such as the NINDS Biomarkers and Human Genetics Repositories, and NeuroBiobank, which integrates brain banking across multiple NIH ICs, will enhance efficiency and empower individual investigators.
Not long ago, neurologists could diagnose diseases of the brain, but do little to treat them. Although medical care is still far from adequate for many neurological disorders, the last two decades have yielded encouraging progress. This includes, for example, more than a dozen drugs for multiple sclerosis, deep brain stimulation therapy for Parkinson’s, and emergency and evidence based preventive interventions for stroke. Previously baffling rare disorders, such as Rett syndrome, spinal muscular atrophy, and muscular dystrophies, whose prospects were especially bleak, are now seeing the first rationally developed therapies entering clinical trials, built on new understanding of the underlying diseases. And astonishing progress is emerging every day from basic research laboratories that bodes well for the future, from nanoscale understanding of how molecules control activity of nerve cells that sense pain and new insights about how dormant genes from viruses may contribute to amyotrophic lateral sclerosis (ALS), to unravelling the mysteries of how the normal brain develops, learns, ignores distractions, and coordinates complex movements. Opportunities for progress against neurological disorders and in promoting optimal brain health throughout life have never been better. NINDS is committed to supporting the highest quality, innovative research by scientists and physicians throughout the United States who can realize this promise.
1JAMA 314:1731-1739 2015
2Neurology Today 14:24-26, 2014
Program Descriptions and Accomplishments:
Overall Budget Policy:
The FY 2017 President’s Budget request is $1,695.180 million, flat over the FY 2016 Enacted level. NINDS emphasizes investigator-initiated research throughout its basic, translational, and clinical programs. Complementing these programs, the Institute solicits research proposals to address unmet mission-critical scientific opportunities, address public health needs, and provide common resources to enhance sharing and enable catalyze productivity. NINDS is a leader in the Neuroscience Blueprint for Neuroscience, which enhances collaboration among the NIH Office of the Director and 15 Institutes and Centers that support neuroscience research. NINDS is also a lead Institute in the development and implementation of the BRAIN Initiative and works closely with other parts of NIH, other Federal agencies, and private groups to implement the scientific vision for the BRAIN Initiative that an interdisciplinary team of scientists developed following extensive consultation with the scientific community. To that end, several BRAIN Initiative notices of funding opportunities have been issued for FY 2016 and others will be issued for FY 2017.
1Circulation 1 34:e6-245, 2013
-->Channels, Synapses, and Circuits:
Ion channels, synapses, and neural circuits are fundamental components of the nervous system. Ion channels control electrical activity of cells. Synapses are specialized connections by which nerve cells influence the activity of other cells. Circuits formed by networks of precisely interconnected nerve cells carry out the information processing that enables us to perceive, think, and act. In addition to basic research on these fundamental components of the nervous system, this program supports studies on dysfunctions of channels, synapses, and circuits, which underlie many neurological disorders. Epilepsy, a prime example, is a major focus of the Channels, Synapses, and Neural Circuits program. NINDS-funded team science projects have identified a variety of new mutations that cause epilepsy and another team is trying to discover the cause of sudden unexpected death in epilepsy (SUDEP). Although research has revealed in remarkable detail the molecular machinery that controls ion channels and synapses, and brain imaging has brought big picture insights about the functions of whole brain regions, technological limitations have held back understanding of how neural circuits work and what goes wrong in disease at that level. The BRAIN Initiative is developing tools to overcome these limitations, building on opportunities from decades of past NIH investments that have been recognized by multiple Nobel Prizes and other awards. Remarkable new methods are now emerging for classifying individual cells, reconstructing the intricate anatomical details of circuits, monitoring activity of thousands of brain cell in real time, and precisely controlling these cells. Hundreds of researchers are already applying these techniques to answer longstanding questions in basic research as well as pain, stroke, epilepsy, Parkinson’s and other diseases.
Budget Policy:
The FY 2017 President’s Budget request is $205.034 million, a decrease of $0.655 million or 0.3 percent less than the FY 2016 Enacted level. In FY 2017, NINDS will continue to balance investigator-initiated research and research targeted to specific mission priorities, including projects funded through the Institute’s translational research and clinical trials programs. Among targeted initiatives, the Epilepsy Centers without Walls (CWOW) bring together the best multidisciplinary teams of investigators, regardless of geographic locations, to focus for multiple years on a specific Benchmarks priority that is better addressed through such a cooperative effort rather than traditional grant mechanisms. Areas of focus have included a very successful neurogenetics CWOW, an active CWOW focusing on Sudden Unexpected Death in Epilepsy (SUDEP), and a new center for FY2016 on finding treatments that modify the disease process itself and combating treatment resistant epilepsy. NINDS and NHLBI have also collaborated to expand the CDC’s Sudden Unexpected Infant Death (SUID) Case Registry to include SUDEP and Sudden Cardiac Death in individuals up to age 24 in 15 states.
Infrastructure, Training programs, and Resources:
The Office of International Activities (OIA), Office of Training, Career Development, and Workforce Diversity, and Office of Clinical Research coordinate activities that span the breadth of NINDS, working with the relevant scientific and disease experts across Institute programs. OIA leads international collaborations that advance the NINDS mission and coordinates the Institute’s global health activities, working closely with the Fogarty International Center. Following the advice of planning panels on workforce diversity, the Institute integrated its programs on diversity within the Office of Training, Career Development, and Workforce Diversity. This office coordinates extensive NINDS extramural programs for training and career development, as well as workforce diversity, tailored to the specific needs of physician-scientists at different stages of their careers. The Office of Clinical Research (OCR) supports infrastructure and programs for clinical research, including early and advanced phase clinical trials and large epidemiological studies, many of which have contributed to the remarkable progress against stroke and made major contributions to understanding health disparities. Over the last several years, OCR has significantly increased the efficiency and effectiveness of large scale clinical trials by enforcing milestones for progress, providing resources to improve patient access and recruitment, developing multi-site clinical networks for stroke, for first in patient studies of new neuro treatments, and for neurological emergency research that provide economies of scale, and pioneering innovations for multi-site trials, such as use of a single Institutional Review Board and master agreements among clinical sites that substantially reduce the time and cost required to launch new trials. The OCR Common Data Elements (CDE) program works with the research and patient advocacy communities to develop standards for specific disorders that facilitate comparison and sharing of clinical data across studies, enhancing the value of these major clinical investments.
Budget Policy: The FY 2017 President’s Budget request is $139.361 million, a decrease of $0.445 million or 0.3 percent less than the FY 2016 Enacted level. The NeuroNEXT clinical network expedites early phase clinical studies through central data and clinical coordinating centers and 25 clinical sites across the U.S. Although it is too early to assess the success of the network, results so far are encouraging, with all four major clinical studies underway having met or on target to reach recruitment goals. In 2017, NINDS will continue to solicit proposals for NeuroNEXT pediatric and adult clinical studies from academic investigators, foundations, small businesses, and industry. The new Stroke Trials Network enhances the effectiveness of small and large stroke clinical trials by eliminating the costs and time associated with developing the infrastructure for each major trial anew and incorporating other efficiencies, such as central Institutional Review Boards and master contract agreements. The Network established regional coordinating centers, a national clinical coordinating center, a national data management center, and clinical sites, and in FY 2015, the first new projects moved forward in the network. In FY 2016 the Institute will continue to seek clinical trial proposals for the Stroke Network from academic, industry, and small business investigators. NINDS is also continuing its support for phase III investigator-initiated clinical trials across all neurological disorders. A full range of NINDS programs in training and career development are also continuing, including individual and institutional grants at the graduate, post-doctoral, and career development levels, and an intensive training course in clinical trials methods for fellows and faculty in the clinical neurosciences. Many of the programs target special needs, such as promoting diversity and enabling neurosurgeons to accommodate research preparation into their demanding training requirements.
Program Portrait: Strategies to Innovate EmeRgeENcy Care Clinical Trials (SIREN)
FY 2016 Level: $1.7 million
FY 2017 Level: $1.7 million
Change: $0.0 million
In FY 2017, NINDS and NHLBI are launching a trans-NIH emergency medicine clinical trials network that will bring together investigators from diverse disciplines to carry out high impact emergency medicine clinical research. The shared infrastructure of the SIREN network, which includes resources of the NCATS Clinical and Translational Science Award program, will improve efficiency and effectiveness of research in the emergency setting. SIREN represents a natural evolution from two previous networks, the NINDS Neurological Emergency Treatment Trials Network (NETT) and NHLBI’s Resuscitation Outcomes Consortium (ROC). A 2015 evaluation of NETT recognized its successes, which include a clinical trial of a rapid treatment for emergency seizures that the Society for Clinical Trials honored as 2014 “trial of the year” in , and highlighted the potential value of expanding its scope. SIREN will build on the NETT experience, with central monitoring, distributed clinical hubs and scalable spokes that expand patient access and trial recruitment. A central Institutional Review Board will further accelerate new trials. The target goal is to complete at least four large, randomized, clinical endpoint trials in pre-hospital and emergency department settings over the first five-year period.
SIREN will address an important public health need and take advantage of synergies between NINDS and NHLBI research programs. There are about 136 million emergency room visits in the United States each year, with a high proportion from people who are disadvantaged minorities. About half of the top 10 complaints relate to the missions of the NINDS or NHLBI, with many areas of intersection between the two institutes, such as neurological outcomes after cardiac or respiratory arrest and hypotensive shock.
Neural Environment:
Non-nerve cells, called glial cells, outnumber nerve cells in the brain. Glial cells, together with specialized blood vessels and immune cells in the brain, maintain the local environment around nerve cells, fight infections, control which molecules enter brain tissue from the circulating blood, and actively shape brain development and synapse function. Neurological disorders can result when non-neuronal cells are compromised; when these cells become aggressors in inflammatory or autoimmune disorders; when cells form tumors; when viruses, bacteria, or parasites infect the nervous system; or when the blood supply to brain cells is compromised to cause stroke. The Neural Environment Program supports basic research on the neural environment, on diseases in which its disruption plays a major role, and on translation of basic scientific knowledge into diagnostic tools, preventive measures, and targeted therapies. The Neural Environment Program coordinates NINDS’ stroke activities, which span several parts of the Institute and have had a major impact on public health. Multiple sclerosis is another common disorder that affects glial cells. Basic research on immunology of the brain and glial cells has fostered private sector development of more than a dozen drugs for multiple sclerosis over the last two decades, and hospitalization rates due to multiple sclerosis have decline by 75 percent over that period. New discoveries on how the brain flushes out potentially harmful substances along blood vessels into the cerebrospinal fluid that surrounds the brain may have implications for many neurodegenerative disorders. Progress against brain tumors, another focus of this program, has been much more difficult to achieve, but a wide range of new studies are revealing potential strategies. Among these, for example, are recent studies on the role of immune system, how cell activity influences tumor growth, better imaging methods to highlight tumor cells, and precision medicine approaches to characterize the molecular changes in individual patient’s tumors to determine the best targeted intervention.
Budget Policy: The FY 2017 President’s Budget request is $241.765 million, a decrease of $0.773 million or 0.3 percent compared to the FY 2016 Enacted level. NINDS will continue to balance investigator-initiated research and research targeted to specific priorities, including research through the Institute’s translational research and clinical trials programs. Expert planning and priority setting panels in stroke research and Alzheimer’s Disease-Related Dementias and well as a 2014 trans-NIH scientific workshop on Small Blood Vessels all emphasized the importance of brain blood vessel health in preventing dementia. NIH is supporting increasing research in this area.
Neurodegeneration:
The Neurodegeneration program focuses on adult onset neurodegenerative diseases, that is, diseases in which brain cells progressively die. Alzheimer’s disease, amyotrophic lateral sclerosis (ALS, or Lou Gehrig’s disease), frontotemporal dementias (FTD), Huntington’s disease, Parkinson’s disease, and vascular cognitive impairment are among the neurodegenerative diseases that affect adults. The National Alzheimer’s Disease Project Act recognized the importance of other types of dementia, such as FTD, vascular dementia, and Lewy body disease, in addition to Alzheimer’s disease. This program supports research to understand all of these diseases and to translate basic findings into clinical practice. Shared mechanisms that contribute to multiple neurodegenerative disorders present a major opportunity for progress. Recent research has revealed, for example, that certain proteins form abnormal aggregates in all of these diseases that may propagate from one brain cell to another, presenting a new target for intervention. Research has also revealed the strong connections between stroke risk factors and dementias, including Alzheimer’s, at every level of investigation from molecular mechanisms to pathology. Over the last several years, the identification of genes that can cause Alzheimer’s disease, Parkinson’s disease, ALS, FTD, and other neurodegenerative diseases has driven progress for both inherited and non-inherited neurodegenerative diseases. Biological pathways revealed by gene identification are often affected in non-inherited diseases as well. In FY 2015, for example, researchers generated mice with mutations that cause human FTD and ALS that will be valuable tools for studying disease mechanisms and potential treatments, and new findings confirmed in multiple laboratories implicate disrupted transport of proteins in an out of the cell nucleus as a major contributor to ALS and potential target for developing drugs.
Budget Policy: The FY 2017 President’s Budget request is $229.267 million, a decrease of $0.733 million or 0.3 percent compared to the FY 2016 Enacted level. NINDS neurodegeneration research will continue to balance investigator-initiated research and solicited research, including therapy development funded through the Institute’s translational research and clinical trials programs. In keeping with the Parkinson’s disease strategic plan, approved by the NANDS Council in 2014, the Institute is maintaining a strong focus on developing biomarkers for this disease that may help predict early onset and track progression to expedite the development of therapies that halt the underlying diseases process. Among other major activities, NINDS has reissued solicitations to continue the Morris K. Udall Parkinson’s Disease Centers of Excellence, which supports a network of centers that work independently and collaboratively, sharing resources and data with the scientific community, to define the causes of and discover improved treatments for Parkinson’s disease. A 2016 Alzheimer’s Disease Related Dementias (ADRD) Summit will update recommendations from a 2013 NINDS led a scientific conference on ADRD’s that were integrated into the National Alzheimer’s Project Act (NAPA) plan, which Congress directed to include the ADRDs. The findings of the summit will guide NIH activities in this area for FY 2017.
Neurogenetics:
Gene defects cause hundreds of rare diseases that affect the nervous system, including ataxias, Down syndrome, dystonias, fragile X syndrome, lysosomal storage diseases, muscular dystrophies, peripheral neuropathies, Rett syndrome, spinal muscular atrophy, Tourette syndrome, and tuberous sclerosis, among many others. Genes also influence susceptibility to common neurological disorders. The Neurogenetics Program supports research on genes that cause or influence neurological disorders, molecular mechanisms through which these genes act, genetic animal and cell models of human disease, and development of treatments for neurogenetic disorders, from the laboratory to the clinic. Basic research to understand how genes and the environment orchestrate brain development is also a key aspect of the neurogenetics program. Over the last two decades, research has identified hundreds of genes related to neurological disorders, which has led to tests that enable physicians to diagnose a disease months or even years more quickly than before, reducing the “diagnostic odyssey” that families who have a child with a rare disease often confront. Gene findings have also led to animal models that mimic key aspects of human diseases, insights about disease mechanisms, and novel strategies for therapy development that target underlying disease mechanisms. Therapies now emerging from this pipeline into clinical testing in the public and private sector have generated cautious optimism that the first effective treatments for spinal muscular atrophy, muscular dystrophies, Rett syndrome, and many other rare genetic diseases may be on the horizon.
Budget Policy: The FY 2017 President’s Budget request is $240.802 million, a decrease of $0.77 million or 0.3 percent compared to the FY 2016 Enacted level. NINDS will continue to support investigator-initiated grants and targeted activities in neurogenetics, including projects funded through the Institute’s translational research and clinical trials programs. The Institute is providing substantial support for the application of whole genome sequencing and other “next generation” genomics methods to neurological disorders. Major continuing trans-NIH programs include the Paul D. Wellstone Muscular Dystrophy Cooperative Research Centers and the Autism Centers for Excellence. In 2015, the Muscular Dystrophy Coordinating Committee approved the updated Action Plan for the Muscular Dystrophies, which will provide guidance not only for NIH activities in muscular dystrophy research, but also for other public and private entities. NINDS will continue to work closely with the NIH Office of Rare Disease Research in supporting and providing disease specific expertise to NINDS mission relevant consortia within the Rare Diseases Clinical Research Network and the Network’s Coordinating Center. NINDS will also continue to support resources for neurogenetics research that enhance the efficiency and effectiveness of research by supporting sharing of data, biological samples, and research tools. In FY2016 NINDS issued a solicitation on clinical trials readiness that will continue in FY2017. The solicitation reflects the growing opportunities in therapy development for rare neurological disorders and will support clinical studies that will fill gaps in the design of upcoming clinical trials, such as validation of outcome measures and biomarkers.
Program Portrait: The NINDS Research Program Award (RPA)
FY 2015 Level: $20.0 million
FY 2016 Level: $20.0 million
Change: $0.0 million
In FY 2016, NINDS launched the Research Program Award (RPA), which will expand in FY 2017. RPA addresses widespread concern that hyper-competition for funding is adversely affecting the efficiency, quality, and innovation of research. Studies suggest that investigators now devote nearly as much time to writing and administering grants as to carrying out research.1 Furthermore, pressure to generate results quickly to renew short-term grants constrains long range, innovative, high risk research.
To address these concerns and increase the value of NINDS funded research, the RPA application, peer review, and award duration differ from traditional R01 grants. The proposal and review accentuate the investigator’s promise and the long-term significance of the proposed research program, rather than the details of specific experiments. RPA is not restricted to senior investigators with a long track record of success. Investigators who are at relatively early stages of their careers (“early established investigators” who have successfully competed for funding) are also eligible. NINDS expects most RPAs to fund an investigator for eight years, with an interim progress evaluation. For investigators with multiple NINDS grants, RPA will combine funding into a single award that sustains the laboratory with up to $750,000 per year direct costs, eliminating the need for multiple applications. However, RPA is also open to outstanding investigators with small laboratories and a single NINDS R01 grant.
RPA represents a new approach to funding neuroscience research, in response to the changing research environment. As for all programs, NINDS will monitor the diversity of applicants and the success of this pilot program to optimize the program and determine what role this strategy should play in the Institute’s overall portfolio in the future.
1Federal Demonstration Partnership 2012 Faculty Workload Survey Research Report (pdf, 2.09 MB)
Repair and Plasticity:
The Repair and Plasticity Program leads NINDS research on traumatic brain injury (TBI), spinal cord injury, and peripheral nerve injury. Research covers the full spectrum, including mechanisms of immediate damage, delayed effects in the hours after initial injury, laboratory development of interventions that minimize damage or stimulate nervous system repair in animal models, and clinical testing in people. The Program also supports fundamental studies of neural plasticity, that is, the ability of the brain and nervous system to change, which has broad implications for recovery following injury or disease. This includes studies of stem cells in the nervous system, including recent studies that point to important role of brain stem cells in normal learning, even in adults. The first clinical trial of stem cell therapy for spinal cord injury is now underway in the private sector, building on years of basic research. The program leads the longstanding NINDS Neural Interfaces Program, which pioneered the entire field of devices that connect to the nervous system to compensate for lost functions, from early development of the first cochlear implants for the hearing impaired to recent brain computer interfaces that enable paralyzed people to control devices with their thoughts. Most recently, decades of investment have let to encouraging recent preliminary results in which electrical stimulation of the spinal cord, combined with intensive training on a treadmill, enabled patients with spinal cord injury to recover some voluntary movement and improve bowel, bladder, and sexual function.
Budget Policy: The FY 2017 President’s Budget request is $142.402 million, a decrease of $0.455 million or 0.3 percent compared to the FY 2016 Enacted level. NINDS continues to balance investigator-initiated research and solicitations, including projects funded through the Institute’s translational research and clinical trials programs. Large observational studies, now underway, will provide critical information to improve clinical care for TBI and clinical trials of interventions. These studies include the Transforming Research and Clinical Knowledge in TBI (TRACK TBI) study of adults and children with TBI at 11 sites in the U.S. and the Multiple Medical Therapies for Pediatric TBI (ADAPT Trial), which is focused on 1000 children with severe TBI. Both of these studies are part of the continuing International TBI Research Initiative in coordination with the European Union and the Canadian Institute of Health Research. These studies use the NINDS TBI Common Data Elements, developed with other Federal agencies and the international research community, and the NIH-Department of Defense led Federal Interagency TBI Informatics System (FITBIR) database to encourage sharing of data. With regard to the important public health issues of sports related TBI, NINDS will continue to work closely with the Foundation for NIH (fNIH) Sports and Heath Research Program. The fNIH created this program with a major donation from the National Football League. The program has launched major cooperative projects to define the scope of long-term changes that occur in the brain years after a head injury or after multiple concussions and to improve diagnostic criteria. In 2017, the Neural Interfaces Program, will continue to solicit and support projects to translate advanced neural prosthetics and other devices up to and through “first in human” clinical demonstrations. NINDS supports device development through a series of notices of funding opportunities coordinated with NINDS Office of Translational Research. The Cooperative Research to Enable and Advance Translational Enterprises – Devices (CREATE-Devices) program provides funding mechanisms tailored to the unique needs of device development at various stages and to the different FDA device approval pathways.
Systems and Cognitive Neuroscience:
Systems of interconnected nerve circuits in the brain, spinal cord, and body control learning, memory, attention, language, thinking, emotion, sensation, movement, and response to pain, as well as sleep, feeding, and drinking. The Systems and Cognitive Neuroscience Program supports research on how the brain carries out these complex functions, on their disruption in neurological disorders, and on promoting recovery. Non-invasive brain imaging is an important research tool for this program, including monitoring of brain activity associated with specific cognitive and behavioral processes. NINDS is a partner in the Human Connectome Project, which has provided unparalleled data for neuroscientists on how parts of the brain interact with one another. The detailed maps of the brain circuits of more than 1,000 people are already producing insights, from unexpected geometric regularities in how nerve fibers are routed, to recent demonstration that connectivity is sufficiently unique to “fingerprint” a person’s brain. Scientists are now beginning to build on this foundation of normal brain connectomes to examine how connectivity is altered during disease. Chronic pain disorders, including migraine and other headaches, are among the most prevalent of all medical conditions and are a high priority for this program. NINDS leads NIH pain research, which is coordinated through the Office of Pain Policy and the NIH Pain Consortium. Notable recent advances in understanding pain include a path-breaking application of a technique called cryo electron microscopy to discern the detailed structure of key molecules that carry pain signals and insights about role of pain receptors in asthma. NINDS also leads the Interagency Pain Research Coordinating Committee (IPRCC), which coordinates the wider Federal and private sector communities. NINDS Office of Pain Policy manages the Interagency Pain Research Portfolio, a database launched in 2014 that provides the public and the research community with information on the breadth and details of pain research and training activities supported by six Federal agencies.
Budget Policy: The FY 2017 President’s Budget request is $201.629 million, a decrease of $0.644 million or 0.3 percent compared to the FY 2016 Enacted level. NINDS balances investigator initiated research and solicitations, including projects funded through the Institute’s translational research and clinical trials programs. Pain continues to be a major area of emphasis, with NINDS coordinating NIH activities through the NIH Pain Consortium. The NINDS Office of Pain Policy, acting through the IPRCC is leading the development of a federal pain research portfolio long term strategy, an effort that is informed by the comprehensive Federal Pain Research Portfolio Analysis. NINDS continues to support the NIH Pain Consortium Centers of Excellence for Pain Education, which act as hubs for the development, evaluation, and distribution of pain management curriculum resources for medical, dental, nursing and pharmacy schools to enhance and improve how health care professionals are taught about pain and its treatment. Among the solicitations continuing in FY2017 are those that focus on chronic overlapping pain conditions, on migraine, and on developing the therapeutic potential of the endocannabinoid system for pain treatment.
Translational Research:
The Office of Translational Research (OTR) leads NINDS preclinical translational activities for all diseases within the Institute’s mission. OTR supports the development of drugs, devices, and biologics, including cell and gene therapies. OTR preclinical therapy development experts work closely with disease-specific experts in extramural programs across the Institute and with the Office of Clinical Research to advance these interventions through the development pipeline to first-in-human studies. NINDS launched the OTR milestone-driven preclinical therapy development program in 2003, and the program has advanced preclinical drug, gene therapy, and cell therapy projects for into further development for Parkinson’s, Batten disease, muscular dystrophy, stroke, and other disorders. In 2015, for example, a small molecule drug for Alzheimer’s disease moved into a phase I clinical trial, a gene therapy for neuropathy showed sufficient progress to receive financing from a private investment firm for further development, and the FDA approved an IND (investigative new drug) application that allowed a biologic therapy for stroke to move into clinical testing. Following extensive analysis, OTR has substantially revised this program. The revised program maintains the milestone-based funding and peer reviewer expertise in therapy development, and adds flexibility to better stage funding to advance projects from early proof-of-concept through first-in-human testing, and to enhance use of expert consultants and contract resources for key steps in the development process that are outside the expertise of most academic laboratories. The program also now tailors funding mechanisms to the different needs of small molecule, device, and biologics therapy development.
Budget Policy: The FY 2017 President’s Budget request is $57.334 million, a decrease of $0.183 million or 0.3 percent compared to the FY 2016 Enacted level. This includes programs led by the Office of Translational Research, but does not include all NINDS translation research activities, which are also supported through budgets of other program areas as appropriate to the disease of focus. The CREATE Bio (Cooperative Research to Enable and Advance Translational Enterprises for Biotechnology Products and Biologics) program supports biotechnology product and biologics therapies, including peptides, proteins, oligonucleotides, gene therapies, and cell therapies. CREATE Devices supports device development along the different FDA-approval pathways for devices. The NIH Blueprint Neurotherapeutics Program, which has been expanded and modified, supports small molecule drug development, with greater flexibility to provide a mix of grant support and access to contract resources. The OTR Innovative Grants to Nurture Initial Translational Efforts (IGNITE) program supports the earlier research and development work necessary to meet the entry requirements for the CREATE and Blueprint Neurotherapeutics therapy development programs. The Anticonvulsant Screening Program, which has contributed to the development of several currently available drugs for epilepsy, will continue, with modifications in response to a comprehensive evaluation by outside experts that was presented to the NANDS Council in 2015. The SBIR and STTR programs are also continuing in FY 2016 under the direction of OTR, including participation in the Commercialization Readiness Pilot program, which was newly authorized in FY2016.
Intramural Research:
IRP conducts research on the NIH campus in Bethesda, Maryland. The Program spans basic and translational neuroscience, neurology, and neurosurgery, and maintains its historical strengths in neuromuscular diseases, neuroimmunology, membrane biochemistry and biophysics, and neurophysiology on the system and synaptic level. The Mark O. Hatfield Clinical Center on the NIH campus in Bethesda, which is a hospital totally dedicated to clinical research, and the Porter Neuroscience Research Center, which integrates neuroscience across NIH ICs and disciplinary boundaries, provide unique resources to the Intramural Research Program. A 2014 Blue Ribbon Panel of distinguished extramural scientists reviewed the NINDS Intramural Research Program. The panel noted that IRP has outstanding leadership, leads NIH in integrating science within and across NIH ICs, provides outstanding support for basic science in the context of the clinical mission, and is vigilant for opportunities to strengthen and expand that clinical mission. The panel also highlighted several areas of special scientific strength ranging from basic molecular neuroscience to imaging research, and specific clinical focus areas, such as neurogenetics, neuroimmunology, neurovirology, movement disorders, stroke, and surgical neurology. Clinical intramural investigators are carrying out innovative therapeutic studies, notably gene therapy studies in patients with Parkinson's disease and Giant Axonal Neuropathy and studies of dormant viral genes in ALS.
Budget Policy: The FY 2017 President’s Budget request is $169.196 million, an increase of $3.318 million or 2.0 percent compared to the FY 2016 Enacted level. The recommendations of the Blue Ribbon Panel and the IRP strategic plan and long term vision will guide the IRP in 2016 as it maintains its strengths in basic and translational neuroscience and recruits exceptional physician scientists to complement its strengths in clinical neuroscience. Based upon the recommendations of the blue ribbon panel the IRP is seeking additional physician scientists in areas including neurogenetics, neuroimmunology and neurovirology, stroke, movement disorders, and surgical neurology. A goal will be to develop a central infrastructure to become a premier site for first-in-human trials of treatments for neurological disorders and stroke, which will require enhancing collaborations with neighboring hospitals and medical schools. The IRP will continue its collaborations through Center for Neuroscience and Regenerative Medicine (CNRM) with the Department of Defense, including the Walter Reed National Military Medical Center and the Uniformed Services University. CNRM, brings together clinicians and scientists across disciplines to catalyze innovative approaches to traumatic brain injury research.
Research Management and Support (RMS):
RMS activities provide administrative, budgetary, logistical, and scientific support in the review, award, and monitoring of research grants, training awards, and research and development contracts. RMS functions also encompass strategic planning, coordination, and evaluation of the Institute’s programs, regulatory compliance, international coordination, and liaison with other Federal agencies, Congress, and the public.
Budget Policy: The FY 2017 President’s Budget request is $68.391 million, an increase of $1.341 million or 2.0 percent compared to the FY 2016 Enacted level.
Budget Authority by Object Class 1
(Dollars in Thousands)
| FY 2016 Enacted |
FY 2017 President's Budget2 |
FY 2017 +/- FY 2016 |
|
|---|---|---|---|
| Total compensable workyears: | |||
| Full-time employment | 522 | 522 | 0 |
| Full-time equivalent of overtime and holiday hours | 0 | 0 | 0 |
| Average ES salary (in whole dollars) | $183 | $183 | $0 |
| Average GM/GS grade | 13.6 | 13.6 | 0.0 |
| Average GM/GS salary (in whole dollars) | $109 | $109 | $0 |
| Average salary, grade established by act of July 1, 1944 (42 U.S.C. 207) (in whole dollars) |
$0 | $0 | $0 |
| Average salary of ungraded positions (in whole dollars) | $0 | $0 | $0 |
| OBJECT CLASSES | FY 2016 Enacted |
FY 2017 President's Budget2 |
FY 2017 +/- FY 2016 |
|---|---|---|---|
| Personnel Compensation: | |||
| 11.1 Full-time permanent | $30,934 | $31,170 | $236 |
| 11.3 Other than full-time permanent | 21,371 | 21,533 | 163 |
| 11.5 Other personnel compensation | 1,309 | 1,319 | 10 |
| 11.7 Military personnel | 790 | 796 | 6 |
| 11.8 Special personnel services payments | 8,841 | 8,908 | 67 |
| Total, Personnel Compensation | $63,244 | $63,726 | $482 |
| 12.0 Personnel benefits | $18,876 | $19,250 | $374 |
| 12.2 Military personnel benefits | 510 | 514 | 4 |
| 13.0 Benefits for former personnel | 0 | 0 | 0 |
| Subtotal, Pay Costs | $82,630 | $83,489 | $859 |
| 21.0 Travel and transportation of persons | $3,896 | $3,896 | $0 |
| 22.0 Transportation of things | 211 | 211 | 0 |
| 23.1 Rental payments to GSA | 1 | 1 | 0 |
| 23.2 Rental payments to others | 55 | 55 | 0 |
| 23.3 Communications, utilities and miscellaneous charges | 808 | 808 | 0 |
| 24.0 Printing and reproduction | 0 | 0 | 0 |
| 25.1 Consulting services | $2,922 | $2,922 | $0 |
| 25.2 Other services | 35,579 | 36,129 | 550 |
| 25.3 Purchase of goods and services from government accounts | 142,448 | 147,378 | 4,929 |
| 25.4 Operation and maintenance of facilities | $2,496 | $2,496 | 0 |
| 25.5 Research and development contracts | 21,574 | 21,547 | -26 |
| 25.6 Medical care | 350 | 350 | 0 |
| 25.7 Operation and maintenance of equipment | 3,359 | 3,359 | 0 |
| 25.8 Subsistence and support of persons | 0 | 0 | 0 |
| 25.0 Subtotal, Other Contractual Services | $208,728 | $214,181 | $5,453 |
| 26.0 Supplies and materials | $10,466 | $10,378 | -$87 |
| 31.0 Equipment | 11,798 | 11,798 | 0 |
| 32.0 Land and structures | 0 | 0 | 0 |
| 33.0 Investments and loans | 0 | 0 | 0 |
| 41.0 Grants, subsidies and contributions | 1,376,585 | 1,370,360 | -6,225 |
| 42.0 Insurance claims and indemnities | 0 | 0 | 0 |
| 43.0 Interest and dividends | 2 | 2 | 0 |
| 44.0 Refunds | 0 | 0 | 0 |
| Subtotal, Non-Pay Costs | $1,612,550 | $1,611,691 | -$859 |
| Total Budget Authority by Object Class | $1,695,180 | $1,695,180 | $0 |
1Includes FTEs whose payroll obligations are supported by the NIH Common Fund.
2Includes mandatory financing.
Salaries and Expenses
(Dollars in Thousands)
| OBJECT CLASSES | FY 2016 Enacted |
FY 2017 President's Budget |
FY 2017 +/- FY 2016 |
|---|---|---|---|
| Personnel Compensation: | |||
| Full-Time Permanent (11.1) | $30,934 | $31,170 | $236 |
| Other Than Full-Time Permanent (11.3) | 21,371 | 21,533 | 163 |
| Other Personnel Compensation (11.5) | 1,309 | 1,319 | 10 |
| Military Personnel (11.7) | 790 | 796 | 6 |
| Special Personnel Services Payments (11.8) | 8,841 | 8,908 | 67 |
| Subtotal Personnel Compensation (11.9) | $63,244 | $63,726 | $482 |
| Civilian Personnel Benefits (12.1) | $17,173 | $19,250 | $374 |
| Military Personnel Benefits (12.2) | 510 | 514 | 4 |
| Benefits to Former Personnel (13.0) | 0 | 0 | 0 |
| Subtotal Pay Costs | $82,630 | $83,489 | $859 |
| Travel & Transportation of Persons (21.0) | $3,896 | $3,896 | $0 |
| Transportation of Things (22.0) | 211 | 211 | 0 |
| Rental Payments to Others (23.2) | 55 | 55 | 0 |
| Communications, Utilities and Misc. Charges (23.3) | 808 | 808 | 0 |
| Printing and Reproduction (24.0) | 0 | 0 | 0 |
| Other Contractual Services: | |||
| Consultant Services (25.1) | 2,922 | 2,922 | 0 |
| Other Services (25.2) | 35,579 | 36,129 | 550 |
| Purchases from government accounts (25.3) | 79,389 | 83,052 | 3,663 |
| Operation and Maintenance of Facilities (25.4) | 2,496 | 2,496 | 0 |
| Operation and Maintenance of Equipment (25.7) | 3,359 | 3,359 | 0 |
| Subsistence and Support of Persons (25.8) | 0 | 0 | 0 |
| Subtotal Other Contractual Services | $185,271 | $188,004 | $4,213 |
| Supplies and Materials (26.0) | $10,466 | $10,378 | -$87 |
| Subtotal Non-Pay Costs | $139,182 | $143,307 | $4,125 |
| Total Administrative Costs | $221,812 | $226,796 | $4,985 |
Details of Full-Time Equivalent Employment (FTE's)
| OFFICE/DIVISION | FY 2015 Actual |
FY 2016 Est. |
FY 2017 Est. |
||||||
|---|---|---|---|---|---|---|---|---|---|
| Civilian | Military | Total | Civilian | Military | Total | Civilian | Military | Total | |
| Office of the Director | |||||||||
| Direct: | 52 | - | 52 | 54 | - | 54 | - | - | 54 |
| Reimbursable: | 1 | - | 1 | 1 | - | 1 | - | - | 1 |
| Total: | 53 | - | 53 | 55 | - | 55 | - | - | 55 |
| Division of Extramural Research | |||||||||
| Direct: | 99 | - | 99 | 100 | - | 100 | 100 | - | 100 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 99 | - | 99 | 100 | - | 100 | 100 | - | 100 |
| Division of Intramural Research | |||||||||
| Direct: | 310 | 9 | 319 | 310 | 9 | 319 | 310 | 9 | 319 |
| Reimbursable: | 6 | - | 6 | 6 | - | 6 | 6 | - | 6 |
| Total: | 316 | 9 | 325 | 316 | 9 | 325 | 316 | 9 | 325 |
| Office of Clinical Research | |||||||||
| Direct: | 15 | - | 15 | 15 | - | 15 | 15 | - | 15 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 15 | - | 15 | 15 | - | 15 | 15 | - | 15 |
| Office of Scientific Liaison | |||||||||
| Direct: | 1 | - | 1 | 3 | - | 3 | 3 | - | 3 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 1 | - | 1 | 3 | - | 3 | 3 | - | 3 |
| Office of Special Programs in Diversity | |||||||||
| Direct: | - | - | - | - | - | - | - | - | - |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | - | - | - | - | - | - | - | - | - |
| Office of Training, Career Development and Workforce Diversity | |||||||||
| Direct: | 5 | - | 5 | 5 | - | 5 | 5 | - | 5 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 5 | - | 5 | 5 | - | 5 | 5 | - | 5 |
| Office of Translational Research | |||||||||
| Direct: | 19 | - | 19 | 19 | - | 19 | 19 | - | 19 |
| Reimbursable: | - | - | - | - | - | - | - | - | - |
| Total: | 19 | - | 19 | 19 | - | 19 | 19 | - | 19 |
| Total | 508 | 9 | 517 | 513 | 9 | 522 | 513 | 9 | 522 |
Includes FTEs whose payroll obligations are supported by the NIH Common Fund.
FTEs supported by funds from Cooperative Research and Development Agreements.
| FISCAL YEAR | Average GS Grade |
|---|---|
| 2013 | 13.4 |
| 2014 | 12.8 |
| 2015 | 13.6 |
| 2016 | 13.6 |
| 2017 | 13.6 |
Detail of Positions
(Dollars in Thousands)
| GRADE | FY 2015 Actual |
FY 2016 Enacted |
FY 2017 President's Budget |
|---|---|---|---|
| Total, ES Positions | 1 | 1 | 1 |
| Total, ES Salary | 183,000 | 183,000 | 183,000 |
| GM/GS-15 | 41 | 44 | 44 |
| GM/GS-14 | 51 | 51 | 51 |
| GM/GS-13 | 80 | 84 | 84 |
| GS-12 | 46 | 45 | 45 |
| GS-11 | 39 | 38 | 38 |
| GS-10 | 4 | 4 | 4 |
| GS-9 | 21 | 21 | 21 |
| GS-8 | 14 | 14 | 14 |
| GS-7 | 8 | 8 | 8 |
| GS-6 | 2 | 2 | 2 |
| GS-5 | 0 | 0 | 2 |
| GS-4 | 3 | 3 | 3 |
| GS-3 | 0 | 0 | 0 |
| GS-2 | 0 | 0 | 0 |
| GS-1 | 0 | 0 | 0 |
| Subtotal | 309 | 314 | 314 |
| Grades established by Act of July 1, 1944 (42 U.S.C. 207): | 0 | 0 | 0 |
| Assistant Surgeon General | 0 | 0 | 0 |
| Director Grade | 3 | 3 | 3 |
| Senior Grade | 0 | 0 | 0 |
| Full Grade | 1 | 1 | 1 |
| Senior Assistant Grade | 1 | 1 | 1 |
| Assistant Grade | 0 | 0 | 0 |
| Subtotal | 5 | 5 | 5 |
| Ungraded | 0 | 0 | 0 |
| Total permanent positions | 314 | 319 | 319 |
| Total positions, end of year | 531 | 534 | 534 |
| Total full-time equivalent (F T E) employment, end of year | 517 | 522 | 522 |
| Average ES salary | 183,00 | 183,000 | 183,000 |
| Average GM/GS grade | 13.6 | 13.6 | 13.6 |
| Average GM/GS salary | 109,176 | 109,176 | 109,176 |
Includes FTEs whose payroll obligations are supported by the NIH Common Fund.
