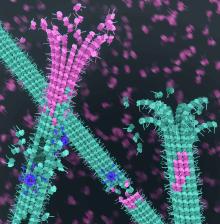
NIH scientists showed that genes linked to spastic paraplegia and microcephaly may normally help strengthen softs spots in microtubules, tiny cylinders that act as internal highways in neurons. Courtesy of Roll-Mecak lab, NIH/NINDS & NHLBI.
NIH study offers new clues to long sought mystery behind disease causing mutations
For years researchers were puzzled by two genes linked to the neurological disorders spastic paraplegia and microcephaly. They thought that the genes severed microtubules, tiny cylinders that give cells their shapes and act as internal highways, but they couldn’t understand why disease-causing mutations destroyed nerves. In a study published in August in Science, NIH scientists suggested that this may be because the proteins that the genes produce, spastin and katanin, repair and reinforce microtubules.
“Cells, like cities, bustle with activity. Cargo and information are transported on a dense and intricate cellular highway system which is made of microtubules. Nerve cells have an especially intricate and long-distance system that allows them to transport cargo and information from one nerve cell to another. That is how we can walk, wiggle our toes or scratch our noses” said Antonina Roll-Mecak, Ph.D., senior investigator at the NIH’s National Institute of Neurological Disorders and Stroke and the senior author of the study. “We found that spastin and katanin actually stabilize soft spots in microtubules. This strengthens the tubules allowing them to grow longer or start new branches.”
Mutations in spastin are the most frequent cause of hereditary spastic paraplegia, a disorder that causes muscle weakness in the legs and feet. Katanin mutations can cause microcephaly, a condition found in children characterized by abnormally small heads and a range of clinical problems, including seizures and problems with thinking, coordination, and balance
Every cell has microtubules, which can be rapidly assembled out of repeating cylindrical arrays of building block proteins, called tubulin. Traditionally it was thought that spastin and katanin worked like pac-men to chomp away, block by block, unneeded microtubule ends. However, this view could not explain why disease-causing mutations, which turned the spastin gene off, eroded microtubules found in axons, the long, thin fibers nerves used to transmit messages to other cells.
Challenging this long-held model, Dr. Roll-Mecak’s team used high powered microscopes to take snapshots of spastin and katanin in action and found that the proteins can strengthen microtubule cylinders. Specifically, their results suggested that spastin and katanin removed older and weaker building blocks, called GDP-tubulin, from the middle of the cylinders. This allowed the spaces to be filled by more stable GTP-tubulin proteins.
“We were completely surprised. We found that these proteins can take little bites out of the microtubules. This allows cells to patch them with a fresh, sturdier type of tubulin building block,” said Dr. Roll-Mecak. “This was unexpected since spastin and katanin were thought to destroy microtubules and the prevailing view in the field had been that microtubules are remodeled only from the ends and not repaired throughout their lengths. This helps explain how disease-causing mutations produce damage to the microtubule highway system in nerves.”
Further experiments suggested that the newly inserted GTP-tubulin not only acted like a speed bump to slow the rate at which microtubules unravel, but also became a launching pad for new microtubule cylinders to grow.
“Our results suggest that spastin and katanin create chances for GTP-tubulin to quickly replace GDP-tubulin, stabilizing microtubules and allowing them to grow,” said Dr. Roll-Mecak. “In the future we plan to explore how nerve cells target and regulate these proteins to remodel their microtubule highways in response to the everchanging needs of the cell.”
Article:
Vemu, A, Szczesna, E.; “Severing enzymes amplify microtubule arrays through lattice GTP-tubulin incorporation”, August 24, 2018, Science; DOI: 10.1126/science.aau1504
This study was supported by Intramural research program at the NIH’s NINDS and National Heart Lung & Blood Institute, grants from the NIH (GM121975) and the Howard Hughes Medical Institute.
For more information:
National Institute of Neurological Disorders and Stroke (NINDS)
National Heart, Lung, and Blood Institute
National Institute of General Medical Sciences (NIGMS)
NINDS Division of Intramural Research
NHLBI Division of Intramural Research
NIH Intramural Research Program (IRP)
###
NINDS (http://www.ninds.nih.gov) is the nation’s leading funder of research on the brain and nervous system. The mission of NINDS is to seek fundamental knowledge about the brain and nervous system and to use that knowledge to reduce the burden of neurological disease.
Part of the National Institutes of Health, the National Heart, Lung, and Blood Institute (NHLBI) plans, conducts, and supports research related to the causes, prevention, diagnosis, and treatment of heart, blood vessel, lung, and blood diseases; and sleep disorders. The Institute also administers national health education campaigns on women and heart disease, healthy weight for children, and other topics. NHLBI press releases and other materials are available online at http://www.nhlbi.nih.gov.
NIGMS is a part of the National Institutes of Health that supports basic research to increase our understanding of biological processes and lay the foundation for advances in disease diagnosis, treatment, and prevention. For more information on the Institute's research and training programs, visit www.nigms.nih.gov.
About the National Institutes of Health (NIH): NIH, the nation's medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit http://www.nih.gov.
