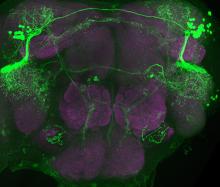
Researchers used state-of-the-art technology to identify how small tweaks in a fruit fly brain circuit can lead to changes in complex behavior. Image courtesy of the Ruta lab, Rockefeller University.
NIH-funded study suggests that slight changes in a brain’s wiring can greatly change behavior
According to a new National Institutes of Health-funded study, it is not destiny that brings two fruit flies together, but an evolutionary matchmaker of sorts that made tiny adjustments to their brains’ mating circuits, so they would be attracted to one another while rejecting advances from other, even closely-related, species. The results, published in Nature, may help explain how a specific female scent triggers completely different responses in different male flies.
“This study reveals how a very small tweak in brain wiring can result in large changes in very complex social behaviors, which can ultimately determine the fate of a species,” said Jim Gnadt, Ph.D., program director at the NIH’s National Institute of Neurological Disorders and Stroke (NINDS), which supported the study. “Understanding how variation in brain circuits leads to changes in behavior is one of the primary goals of the NIH’s BRAIN Initiative and this study provides a piece of the puzzle.”
Vanessa Ruta, Ph.D., professor at Rockefeller University in New York City, and her colleagues used cutting-edge genetic tools to compare the brain circuits behind courtship behavior in two closely related species of fruit fly, D. melanogaster and D. simulans.
Previous studies showed that although males from both species could detect a specific pheromone, or scent, called 7,11-heptacosadiene (7,11-HD), their reactions to it were very different. Male D. melanogaster flies found it attractive while D. simulans males avoided females that carried it. In this study, Dr. Ruta and her team discovered that slight differences in the way the fly’s brains are wired may control these opposite reactions.
“From a fly’s perspective, courtship is the most important decision it will make, and so evolutionary processes have really fine-tuned their brains to ensure optimal behaviors, leading them to their ideal mates,” said Dr. Ruta.
In fruit flies, pheromones are detected by sensory neurons on the legs, and that information travels to P1 neurons that sit in the lateral protocerebral complex (LPC) in the brain. In between the sensory and P1 neurons, the wiring splits so that the P1 cells can be either turned on or off.
P1 neurons trigger courtship activity in D. melanogaster males but have not been studied in other fly species. When the P1 neurons were turned on in D. simulans males, they tried mating with nearby objects including unsuitable targets such as females from different species as well as rotating magnets. This suggests that P1 neurons may be important for sparking courtship behaviors across species.
Dr. Ruta and her team used state-of-the-art technology to watch brain cells light up in real time as male flies were exposed to D. melanogaster and D. simulans females releasing 7,11-HD. When D. melanogaster males were exposed to females from their species, there was a lot of activity by the P1 neurons. Exposure to D. simulans females did not turn on those neurons. However, P1 neurons in the D. simulans males did not light up when they were exposed to females from their species, suggesting that differences in P1 neurons may underlie species-specific responses to 7,11-HD.
Additional experiments suggested that as pheromone signals traveled from the legs to P1 neurons, that information was conveyed differently in the two species of fruit fly by brain cells that communicate directly with P1 neurons. Specifically, P1 neurons appeared to receive lots of excitation in D. melanogaster flies but got more calming signals in D. simulans flies, which led to opposite responses when the animals were exposed to 7,11-HD. These findings also suggested that in some species of flies, absence of pheromone signaling may be more attractive than the presence of certain scents.
Future experiments will look at similar behaviors in other species of fruit flies. More research is needed to learn the mechanisms that drive evolutionary changes in brain circuitry, which may have important consequences for individual and social behavior.
This study was supported by NINDS (NS0879422013), New York Stem Cell Foundation, the Pew Foundation, the McKnight Foundation, the Irma T. Hirschl Foundation, the Alfred P. Sloan Foundation, National Science Foundation and Kavli Foundation.
For more information:
National Institute of Neurological Disorders and Stroke
The BRAIN Initiative
Reference:
Seeholzer LF. et al. Evolution of a central neural circuit underlies Drosophila mate preferences. Nature. July 11, 2018. https://www.nature.com/articles/s41586-018-0322-9
This press release describes a basic research finding. Basic research increases our understanding of human behavior and biology, which is foundational to advancing new and better ways to prevent, diagnose, and treat disease. Science is an unpredictable and incremental process—each research advance builds on past discoveries, often in unexpected ways. Most clinical advances would not be possible without the knowledge of fundamental basic research.
###
The NINDS is the nation’s leading funder of research on the brain and nervous system. The mission of NINDS is to seek fundamental knowledge about the brain and nervous system and to use that knowledge to reduce the burden of neurological disease.
About the National Institutes of Health (NIH): NIH, the nation's medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit https://www.nih.gov.
